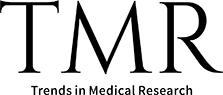Long COVID: Key Features and Current Trends in Management
| Received 20 Jan, 2026 |
Accepted 16 Mar, 2026 |
Published 20 Mar, 2026 |
Long COVID, or Post-Acute Sequelae of COVID-19 (PASC), has emerged as a major public health challenge, affecting a substantial minority of people months to years after the acute illness. Recent cohort studies and meta-analyses suggest that 10-30% of infected individuals report persistent symptoms, with some series indicating that ~45% still experience complaints four months after infection, and higher prevalence in lower- and middle-income settings. Clinically, Long COVID is heterogeneous, with cardinal manifestations including fatigue, exertional intolerance, dyspnea, chest pain, dysautonomia, sleep disturbance, cognitive impairment (“brain fog”), headache, myalgia, and a range of neuropsychiatric, cardiovascular and gastrointestinal symptoms. Reported risk factors include female sex, higher Body Mass Index (BMI), smoking, comorbidities such as diabetes and hypertension, severe acute COVID-19, prolonged hospitalization or ICU stay, and a high burden of symptoms during the acute phase. Pathogenesis is likely multifactorial, involving viral persistence, immune dysregulation, autoimmunity, endothelial dysfunction, dysautonomia, and mitochondrial and metabolic dysfunction affecting multiple organ systems. Management is therefore complex and symptom-directed, emphasizing careful phenotyping, rehabilitation, psychological support, and secondary prevention of complications. The Johns Hopkins Post-Acute COVID-19 Team (PACT) at the Bayview Medical Center in Baltimore provides a model of multidisciplinary care: a collaborative, ambulatory program led by pulmonary and critical care medicine and physical medicine and rehabilitation, and integrating neurology, psychiatry, pharmacy, primary care, and other subspecialties. Using a multi-pronged approach that combines standardized assessment, tailored pharmacologic and non-pharmacologic interventions, structured rehabilitation, and coordinated follow-up, PACT addresses clusters of respiratory, neurocognitive, mental health and functional impairments in a single care pathway. This model illustrates how interdisciplinary, post-acute clinics can respond to the complex needs of long COVID “long-haulers”, while simultaneously generating data to refine definitions, clarify mechanisms and improve evidence-based treatment.
| Copyright © 2026 Kaushik Bharati. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Coronavirus Disease 2019 (COVID-19), caused by the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), has produced not only waves of acute infection but also a substantial burden of persistent, often disabling sequelae now termed Long COVID, post-COVID conditions (PCC), or Post-Acute Sequelae of COVID-19 (PASC). Long COVID is increasingly conceptualised as an infection-associated chronic condition in which new, recurrent, or ongoing symptoms are present for ≥3 months after acute infection, in a continuous, relapsing-remitting, or progressive pattern involving one or more organ systems.
|
Since patient communities first popularised the term “long-haulers” in 2020, observational cohorts, population-based surveys, and systematic reviews have confirmed that Long COVID affects a sizeable minority of those infected. Estimates of prevalence vary with case definition, follow-up duration, and study design, but pooled analyses suggest that roughly 10-30% of adults experience at least one persistent symptom beyond 4-12 weeks, with higher proportions in hospitalised cohorts and in some low- and middle-income countries (LMICs). Symptoms may persist for up to four years in a subset of survivors, compared to one month in the case of the acute phase of COVID-19. This can produce measurable impacts on function, employment and quality of life (Fig. 1).
Clinically, Long COVID is heterogeneous and multi-systemic. Large series describe fatigue, post-exertional symptom exacerbation, dyspnea, chest pain, cough, palpitations, dysautonomia, sleep disturbance, cognitive impairment (“brain fog”), headache, myalgia, arthralgia, anosmia/dysgeusia, mood and anxiety symptoms, and a variety of gastrointestinal, dermatologic, and menstrual disturbances as dominant complaints. These manifestations cluster into partially overlapping phenotypes involving respiratory, neurocognitive, cardiovascular, autonomic and mental health domains, and often fluctuate over time.
Several demographic and clinical risk factors for Long COVID are now recognised. Systematic reviews and meta-analyses indicate that female sex, older age, higher Body Mass Index (BMI), smoking, and comorbidities such as diabetes, cardiovascular disease, chronic lung disease, chronic kidney disease, and anxiety or depression are associated with increased risk1.
The pathogenesis of Long COVID is complex and likely multifactorial. Emerging mechanistic work implicates persistent viral reservoirs or delayed viral clearance, chronic immune activation, autoimmunity, and endothelial injury with associated microvascular thrombosis and “microclots”. Additional contributors include dysautonomia, mitochondrial and metabolic dysfunction, coagulation complement pathway activation, and disruption of the gut-lung and gut-brain axes. These mechanisms can produce sustained multi-organ injury, including the pulmonary, cardiovascular, neurological, endocrine, and renal systems.
Given this heterogeneity, management of Long COVID is inherently challenging. High-quality randomised trials of targeted therapies remain limited, and most current care is symptom-directed and pragmatic, combining pharmacological treatment of organ-specific complications with rehabilitation, self-management support, and attention to mental health. National and international guidelines increasingly emphasise comprehensive assessment, validation of patients’ experiences, screening for red-flag conditions, and longitudinal follow-up, ideally delivered through coordinated, multi-disciplinary services2. In response, post-COVID clinics have been rapidly established worldwide. One of the earliest and most influential models is the Johns Hopkins Model, present at the Johns Hopkins Bayview Medical Center in Baltimore, United States.
|
As the pandemic transitions into an endemic phase, the cumulative burden of Long COVID continues to grow, particularly in health systems with constrained resources. A nuanced understanding of its emergence, epidemiology, clinical spectrum, risk factors, pathogenesis, and management approaches is essential for clinicians, researchers, and policymakers. This review will synthesise the evolving evidence on Long COVID, highlight current gaps in knowledge, and use the Johns Hopkins PACT programme as a case study of a multi-disciplinary, multi-pronged model of care for affected patients.
EPIDEMIOLOGY
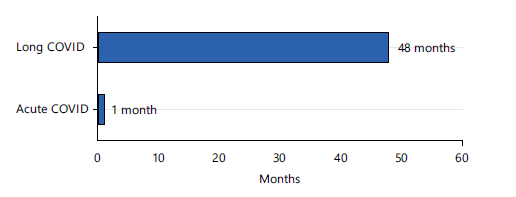
Based on the WHO-Delphi clinical case definition, Long COVID is characterized by symptoms that arise after confirmed or probable SARS-CoV-2 infection, persist for ≥12 weeks, and cannot be explained by alternative diagnoses. Recent population-based cohorts, meta-analyses, and global modeling studies converge on the conclusion that a substantial proportion of people develop long-lasting sequelae after acute infection. Global estimates from the Global Burden of Disease Long COVID Collaborators suggest that about 6.2% of symptomatic infections in 2020-2021 resulted in at least one of three canonical symptom clusters (persistent fatigue with pain/mood changes, ongoing respiratory symptoms, or cognitive problems) at 3 months, corresponding to tens of millions of incident cases in just the first two pandemic years3. The global distribution of Long COVID is presented in Fig. 2.
Long COVID prevalence remains high even when restricted to symptoms persisting ≥12 weeks. A global meta-analysis showed that 43% of individuals had ongoing symptoms ≥28 days after infection, with higher prevalence in hospitalized than in non-hospitalized cohorts4. A study that analyzed data from 194 cohorts found that approximately 45% of survivors reported at least one unresolved symptom at a mean of four months, with many still symptomatic at 12 months5.
Symptomatically, Long COVID is a multisystem condition dominated by fatigue, breathlessness, and cognitive dysfunction. Rapid reviews and meta-analyses document a wide constellation of complaints affecting almost every organ system, but consistently identify fatigue, exercise intolerance, dyspnea, chest pain, palpitations, “brain fog”, headache, sleep disturbance, anxiety, and depressive symptoms among the most frequent. These symptom clusters translate into substantial disability: Long-COVID cohorts report reduced quality of life, impaired activities of daily living, and work loss months after infection, highlighting that epidemiologic burden is driven predominantly by morbidity rather than mortality.
|
Risk is not evenly distributed. Narrative reviews emphasise that age modifies phenotype as well as risk, with older adults experiencing more cardiometabolic and frailty-related outcomes, whereas younger adults often report prominent dysautonomia, fatigue and neurocognitive complaints. Notably, children generally have lower absolute risk, but paediatric cohorts still show non-trivial rates of persistent symptoms, underscoring the intergenerational reach of the condition.
Mortality directly attributed to post-COVID-19 condition is comparatively rare but increasingly recognised in vital-registration systems. A national analysis from Italy reported a mortality rate of 5.1 per 100,000 inhabitants in 2021, with higher rates in men and older adults6. Importantly, multiple large cohorts have shown excess long-term mortality and cardiometabolic morbidity among COVID-19 survivors, suggesting that Long COVID contributes to a broader pattern of increased post-infectious health risks.
Thus, the epidemiologic evidence indicates that Long COVID is a common consequence of SARS-CoV-2 infection, affecting a substantial minority of survivors worldwide and generating a heavy burden of chronic morbidity, disability, health-care utilisation and, to a lesser extent, premature mortality. As such, robust surveillance systems, harmonised case definitions, and high-quality longitudinal cohorts are essential to refine burden estimates, understand risk modifiers and inform prevention and rehabilitation strategies.
TRANSITION FROM ACUTE COVID-19 TO LONG COVID
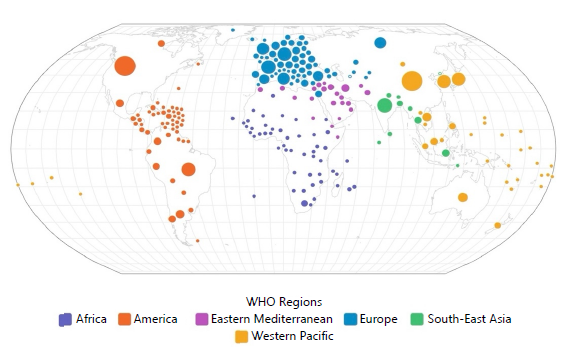
COVID-19 is now recognized as a continuum extending from an acute viral illness into a post-acute convalescent phase and, in a substantial minority, into Long COVID or post-COVID-19 condition. Acute COVID-19 is usually defined as the first four weeks from symptom onset; a post-acute or “ongoing symptomatic” phase spans roughly 4-12 weeks; and Long COVID describes symptoms that persist or recur beyond 12 weeks, generally using a ≥3-month threshold from onset and a duration of at least two months not explained by another diagnosis. This continuous trajectory may extend well beyond 24 weeks in many patients (Fig. 3).
The acute phase is dominated by active SARS-CoV-2 replication in respiratory and other Angiotensin Converting Enzyme 2 (ACE2)-expressing tissues, a brisk innate and early adaptive immune response, endothelial injury and a pro-thrombotic state. Severe disease features hyperinflammation, micro- and macrovascular thrombosis and multi-organ involvement affecting lungs, heart, kidneys and nervous system. Clinically, patients present with fever, cough, dyspnea, myalgia, headache and often anosmia or ageusia. In severe cases they develop pneumonia, ARDS, myocarditis, arrhythmias, acute kidney injury and thromboembolic events. Older age, male sex, obesity, diabetes and cardiovascular disease increase the likelihood of both severe acute outcomes and later persistent symptoms.
From about 2-4 weeks onward, three broad trajectories appear: Complete or near-complete recovery; slow recovery with residual symptoms such as fatigue and exertional dyspnea; or early-onset, multisystem symptoms that never fully remit and evolve into long COVID. In the 4-12 week post-acute window, overt viral replication has usually resolved, but tissue repair, gradual resolution of inflammation and subacute complications continue. Ongoing immune activation at lower levels, consequences of organ damage (for example, lung parenchymal injury, myocarditis, thromboembolism), post-ICU syndrome, deconditioning and possible low-level viral persistence all contribute to a heterogeneous clinical picture. Patients commonly report fatigue, breathlessness, chest tightness, cough, sleep disturbance, anxiety or low mood, anosmia, myalgia and headache. A study confirms a high burden of fatigue, dyspnea, sleep problems, and cognitive complaints at 3-6 months7.
Long COVID or post-COVID-19 condition is defined by WHO as symptoms occurring usually three months from onset, lasting at least two months, and not explained by another diagnosis. They may persist from the acute episode, recur after an asymptomatic period or newly emerge and typically fluctuate. It is characteristically multisystem. Dominant clusters include profound fatigue with post-exertional symptom exacerbation, neurocognitive complaints (“brain fog,” slowed thinking, memory impairment), dysautonomia (orthostatic intolerance, postural tachycardia, palpitations), dyspnea and chest pain, chronic headaches and myalgia, sleep disturbance, and anxiety, depression or PTSD features. At 6-12 months after hospital discharge, persistent fatigue, muscle weakness, sleep difficulties, psychiatric symptoms and reduced exercise capacity have been observed in large proportions of patients, with only gradual improvement. Two-year follow-up studies still find at least one persistent symptom in around 60-70% in some series, with fatigue, pain and memory problems particularly prominent8.
Long COVID appears to share roots with the acute and post-acute phases but with distinct chronic mechanisms. Proposed processes include viral or antigen persistence in tissues, chronic immune dysregulation with altered cytokine profiles and lymphocyte subsets, autoantibody production, endothelial dysfunction and microvascular thrombosis, autonomic imbalance, mitochondrial and metabolic dysfunction, and reactivation of latent pathogens such as Epstein-Barr Virus (EBV). Different patients likely harbour different constellations of these mechanisms, explaining the spectrum of clinical phenotypes and overlaps with myalgic encephalomyelitis/chronic fatigue syndrome, dysautonomia and chronic pain syndromes.
Despite differences in timing and dominant biology, the phases share important continuities. Fatigue, dyspnoea, chest pain, cognitive complaints and sleep disturbance can begin acutely and persist or recur in post-acute and Long COVID. Multisystem involvement is evident from early infection onward, and psychological distress interacts with but does not fully explain the underlying biological disease. Severe acute disease and a higher number of acute symptoms predict greater risk of long-term sequelae, yet Long COVID also affects many non-hospitalised and previously healthy individuals. At the population level, the emergency phase of acute COVID-19 has attenuated, but Long COVID remains an ongoing chronic health burden, with a substantial proportion of infected individuals experiencing persistent symptoms for many months and a minority remaining functionally impaired for several years.
SYMPTOMS
Long COVID (post-COVID-19 condition) is a multisystem disorder with >200 reported symptoms that fluctuate over time and cluster into a few dominant domains: fatigue/post-exertional malaise, respiratory, neurocognitive, autonomic, cardiovascular, pain, sleep and mental-health symptoms, with many others less common.
SARS-CoV-2 spreads throughout the body and infects almost all the organs, as the ACE2 receptor is ubiquitously distributed in human body, resulting in a wide spectrum of symptoms. These are highlighted in Fig. 4.
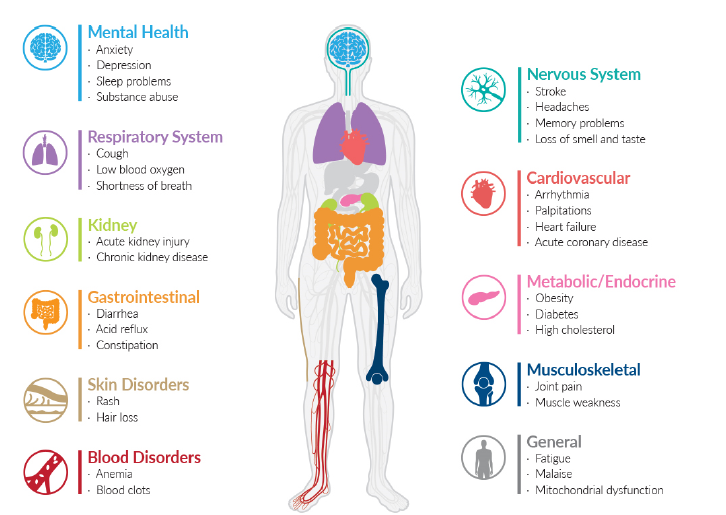
|
GENERAL SYMPTOMS
Fatigue is the most common symptom, affecting 30-60% of patients in the first 6-12 months and persisting in a substantial minority beyond two years. Post-exertional malaise (PEM), with delayed symptom worsening after minor physical or mental effort, overlaps with myalgic encephalomyelitis (ME) or chronic fatigue syndrome (CFS) phenotypes and is reported in around half of patient-led cohorts. Other general features include low-grade fever, night sweats, weight change and global weakness. Additionally, myalgia, arthralgia, muscle weakness and fibromyalgia-like widespread pain are prominent and affect 20-30% of patients at 3-12 months. These symptoms are closely linked to fatigue and PEM and contribute substantially to disability9.
Respiratory symptoms: Breathlessness is a leading long-term symptom, with pooled prevalence around 20-40% at 3-12 months, sometimes associated with reduced diffusing capacity of the lungs for carbon monoxide (DLCO), restrictive patterns or residual imaging abnormalities, and sometimes occurring despite normal tests. Chronic cough, chest tightness and “air hunger” are reported in 10-20% of patients, and may reflect small-airways disease, vascular changes, dysfunctional breathing or dysautonomia10.
Cardiovascular symptoms: Palpitations, chest pain, and exercise intolerance are frequently reported (roughly 10-30%). Large cohort studies show increased 1-year risks of arrhythmias, myocardial infarction, heart failure, and thromboembolic disease even after non-severe infection, indicating a genuine excess cardiovascular burden associated with Long COVID11.
Neurological and cognitive symptoms: Cognitive dysfunction (“brain fog”) with impaired attention, memory and processing speed affects 20-30% of patients in many cohorts and can persist for years. Headache is one of the most frequent symptoms overall and often persists beyond the acute phase. Other neurological features include dizziness, paresthesias, neuropathic pain, tinnitus, sleep-wake disruption and, more rarely, stroke, seizures and peripheral neuropathies. Smell and taste disturbances (anosmia, hyposmia, dysgeusia) remain in around 10-30% of patients in some series12.
Autonomic symptoms: Dysautonomia, including postural orthostatic tachycardia syndrome (POTS), orthostatic hypotension, temperature dysregulation, sweating abnormalities and gastrointestinal or bladder dysmotility, is increasingly recognized as a core component of Long COVID. Overt late dysautonomia has been reported in 2-5% of post-COVID patients, accompanied by autonomic symptoms such as tachycardia, dizziness and cognitive worsening on standing13.
Mental and sleep symptoms: Long COVID is associated with increased rates of anxiety, depression, post-traumatic stress and irritability, with estimates around 20-40% in many studies. Sleep problems like insomnia, non-restorative sleep or hypersomnia occur in 20-30% and interact with fatigue, pain and cognitive difficulty14.
Other symptoms: Persistent diarrhea, abdominal pain, nausea, dyspepsia, bloating and altered bowel habit are described in 5-15% of Long-COVID cohorts and often overlap with autonomic dysfunction and gut microbiome alterations. Post-COVID cohorts also show increased risks of new-onset diabetes, dyslipidemia and other metabolic disorders, which may manifest as polyuria, polydipsia, weight change and fatigue. Thyroid dysfunction and menstrual irregularities have also been reported. Hair loss (Telogen effluvium) is among the most common non-respiratory symptoms, with meta-analysis estimates around 20-25%. Other dermatologic features include rashes, urticaria and chilblain-like lesions. ENT and ophthalmic manifestations such as sore throat, chronic rhinitis, tinnitus, hearing changes, dry eyes and blurred vision are individually less common but clinically relevant.
RISK FACTORS
Major risk factors for developing Long COVID (post-COVID-19 condition) cluster into demographic, clinical and virological domains, with broadly consistent signals across large cohorts and meta-analyses.
Demographic and social factors: Risk rises from young adulthood into mid-life, with several large datasets suggesting the highest risk around 40-69 years rather than the very oldest ages. Long COVID is more prevalent in adults than in children. Being a female is one of the most consistent risk factors compared to men. Ethnic minority status, socioeconomic deprivation and high-exposure occupations (especially healthcare) are also associated with increased risk, reflecting both higher infection exposure and structural factors that influence health and access to diagnosis.
Comorbidities and lifestyle factors: Several chronic conditions modestly but consistently increase the probability of Long COVID. Obesity and higher BMI are repeatedly associated with persistent symptoms. Chronic respiratory disease, particularly asthma and COPD, and cardiometabolic disease, such as diabetes, ischemic heart disease and hypertension have been linked to higher risk. Mental health conditions, especially pre-existing depression and anxiety, also show a consistent association with later Long COVID diagnoses. Immunosuppression and some autoimmune diseases may confer additional risk, though findings are more heterogeneous. Smoking (current or past) is another reported risk factor. Overall, individuals with multiple comorbidities have a substantially higher risk than those without underlying disease.
Prolonged hospitalization: Greater acute illness severity is a strong and reproducible predictor. Hospitalization, particularly ICU admission, mechanical ventilation and prolonged stay, are consistently associated with markedly increased risk of Long COVID. In the N3C/RECOVER cohort, hospitalization nearly quadrupled the chances of receiving a Long COVID diagnosis.
Breakthrough infections: Vaccination before infection appears partially protective. It has been observed that at least two doses of COVID-19 vaccine reduce, but do not abolish the risk of Long COVID due to breakthrough infections. Recent systematic reviews estimate risk reductions on the order of 20-40%, with vaccinated individuals still able to develop post-COVID-19 condition. Several national and global analyses report lower Long COVID prevalence after omicron compared with pre-omicron infections, but with a persisting substantial burden. Reinfection further increases risk, as revealed by longitudinal studies. These suggest that repeated SARS-CoV-2 infections raise the likelihood of developing Long COVID or worsening of existing symptoms, even if earlier infections were uncomplicated.
The risk factors highlighted above are probabilistic, not deterministic. This indicates that many high-risk individuals recover fully, and Long COVID can occur after mild, first-episode infection in previously healthy, vaccinated people. Nonetheless, convergent evidence indicates that mid-life adults, particularly women with obesity, cardiometabolic or respiratory disease, high acute symptom burden or severe initial illness, who are unvaccinated or repeatedly infected, constitute groups at substantially higher risk and may warrant prioritized prevention and follow-up.
PATHOGENESIS
The pathogenesis of Long COVID is increasingly understood as a heterogeneous, multisystem disorder involving overlapping mechanisms rather than a single causal pathway. Persistent viral reservoirs or antigens, chronic immune dysregulation, endothelial dysfunction, autonomic disturbance, mitochondrial impairment, organ-specific injury, microbiome alterations and latent virus reactivation all appear to contribute to the syndrome. These mechanisms likely interact to produce different “endotypes” of Long COVID across individuals and organ systems. The major mechanisms and pathways of the pathogenesis process are highlighted below.
Viral persistence and chronic antigenic stimulation: Evidence of viral persistence has been demonstrated through the detection of SARS-CoV-2 RNA, spike and nucleocapsid proteins in tissues months after acute infection, including gut, brain, heart and lymphoid organs. It has been shown that tissue viral positivity is associated with Long COVID symptoms. Also, persistent infection lasting ≥60 days has been linked to >50% higher odds of Long COVID15. Moreover, it has been proposed that reservoirs in immune-privileged sites such as the gut and CNS may maintain chronic antigenic stimulation, drive inflammation and contribute to T-cell exhaustion and cytokine abnormalities.
Immune dysregulation and autoimmunity: Immune dysregulation is one of the most consistent findings, characterized by persistent elevation of pro-inflammatory cytokines, altered monocyte and NK-cell phenotypes, expanded exhausted CD8+ T-cells and reduced naïve T and B cells. Sustained immunological dysfunction has been reported up to eight months after mild infection. Acute-phase immunologic signatures have also been identified, including autoantibodies and Epstein-Barr Virus (EBV) reactivation, which can predict Long COVID16. Autoimmunity also appears relevant, as functional autoantibodies against G-protein-coupled receptors (GPCRs), antinuclear and antiphospholipid antibodies, and ACE2 autoantibodies have been reported in Long COVID, which may contribute to vascular dysregulation, dysautonomia, and chronic inflammation.
Endothelial dysfunction and coagulopathy: Endothelial dysfunction and coagulopathy are central mechanisms. SARS-CoV-2 infects endothelial cells, producing sustained endothelitis, elevated von Willebrand factor, and a pro-thrombotic state. Persistent endothelial glycocalyx damage, circulating endothelial cells and microvascular dysfunction with raised D-dimer up to one year post-infection have been reported by Wu et al.17. Microclots composed of fibrinoid aggregates resistant to fibrinolysis have been identified in Long COVID plasma and hypothesized to impair microcirculation and oxygen delivery. These vascular abnormalities may underlie exertional intolerance, chest pain, dyspnea, and increased long-term cardiovascular risk.
Autonomic dysfunction: Autonomic dysfunction and neuroinflammation contribute substantially to symptomatology. Dysautonomia, including POTS, orthostatic intolerance, and thermoregulatory instability, has been widely documented. Long COVID patients with autonomic dysfunction were first described in 202118. Mechanisms include direct or immune-mediated injury to brainstem autonomic nuclei, small fiber neuropathy, autoantibodies to adrenergic and muscarinic receptors, and impaired cerebral blood flow driven by endothelial dysfunction and microclots.
Gut microbiome dysbiosis: Gut microbiome alterations are increasingly being recognised. The gut is a reservoir where persistent viral RNA/protein is observed months after infection. Longitudinal studies show dysbiosis correlating with systemic inflammation and symptom persistence, and animal models demonstrate behavioral and metabolic changes following fecal transplantation from post-COVID individuals. Gut barrier dysfunction may permit microbial translocation, fuelling systemic inflammation and affecting the gut-brain axis. Reactivation of latent viruses, particularly EBV, also appears to contribute to the severity of dysbiosis.
Integrative endotypes: Overall, Long COVID likely reflects interacting endotypes, including viral persistence, autoimmunity, endothelial dysfunction, autonomic dysfunction, and metabolic impairment19.
Pathogenesis of Long COVID on the basis of the three major stages of the disease (acute, inflammatory, recovery) is summarized in Fig. 5.
Disease dynamics: The disease dynamics involve three phases, namely, (i) acute phase, characterized by a positive qRT-PCR (quantitative reverse transcription polymerase chain reaction), also known as real-time RT-PCR test, (ii) post-acute phase, characterized by a negative qRT-PCR test, and (iii) Long COVID phase, characterized by persistent or relapsing disease with fluctuating qRT-PCR positivity.
ACUTE PHASE
The disease course of Long COVID begins with the acute phase of SARS-CoV-2 infection, during which patients are typically qRT-PCR positive. Viral load in the upper respiratory tract rises rapidly, often peaking around symptom onset, and then gradually declines over the following 1-2 weeks. Although the infectious virus usually diminishes earlier, qRT-PCR may remain positive for longer periods because it detects viral RNA rather than viable virus. During this phase, SARS-CoV-2 can directly infect epithelial, endothelial, and immune cells, leading to tissue injury and systemic inflammation. Endothelial involvement and microvascular dysfunction are prominent features and contribute to early multi-organ injury involving the lungs, heart, kidneys, brain, and vascular system. The magnitude and duration of viral replication, together with the host immune response, play a key role in determining downstream outcomes20.
Transition to post acute phase: As viral RNA in the upper respiratory tract falls below the detection threshold, most patients transition to a post-acute phase, typically 4-12 weeks after infection, and become qRT-PCR negative on routine testing. Despite apparent virological clearance, many individuals continue to experience symptoms. This phase is characterized less by active viral replication and more by persistent inflammation, immune dysregulation, endothelial injury, and impaired tissue repair. Evidence suggests that delayed viral clearance during the acute phase increases the risk of prolonged symptoms. Importantly, the absence of qRT-PCR positivity does not exclude the presence of viral RNA or antigens in tissue compartments such as the gastrointestinal (GI) tract or lymphoid organs, which may continue to stimulate the immune system21.
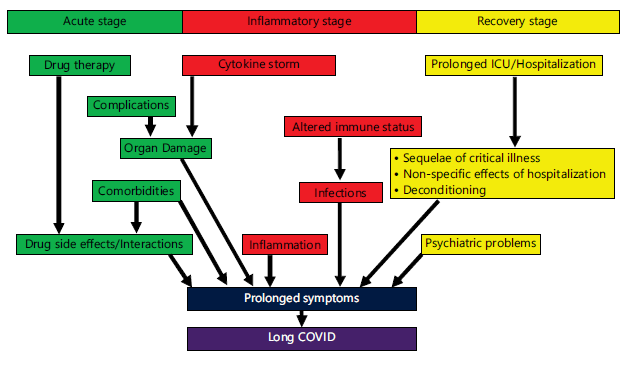
|
Long COVID phase: When symptoms persist or newly emerge beyond 12 weeks, the condition is termed Long COVID. Clinically, this phase is marked by a relapsing-remitting course, with fluctuations in symptom severity. Most patients remain qRT-PCR negative in nasopharyngeal samples; however, some show intermittent or recurrent qRT-PCR positivity. This may reflect low-level RNA shedding, viral rebound after partial suppression, reinfection, or compartmentalized viral persistence rather than a full reactivation resembling acute infection. In such cases, viral loads are usually low, with high cycle-threshold (Ct) values, contrasting with the high viral loads of primary acute disease16.
Viral load dynamics across phases: Across the disease continuum, viral load shows a biphasic or multiphasic pattern. High viral loads dominate the acute phase, followed by apparent clearance in the post-acute phase. In Long COVID, viral RNA is often undetectable in respiratory samples, yet increasing evidence supports the persistence of virus or viral RNA in multiple organs, months after initial infection. This persistent antigenic stimulation may perpetuate immune activation and chronic symptoms even in the absence of sustained respiratory tract infection.
Multi-organ damage and systemic pathophysiology: Long COVID is fundamentally a multi-organ disorder. Pulmonary sequelae include persistent dyspnea, impaired diffusion capacity, and exercise intolerance. Cardiovascular involvement manifests as chest pain, arrhythmias, myocarditis-like syndromes, and postural tachycardia, reflecting endothelial injury and microvascular ischemia. Neurological and autonomic symptoms, commonly described as brain fog, headaches, sleep disturbance, and dysautonomia, are linked to neuroinflammation, vascular injury, and altered neuroimmune signaling. Systemically, persistent endothelial dysfunction, microclot formation, and immune abnormalities, including autoantibody production, act as amplifying mechanisms affecting multiple organs simultaneously.
Thus, the multimodal pattern of Long COVID dynamics reflects complex interactions between viral persistence, host immune responses, and cumulative organ damage22.
Rehabilitation and supportive care: Currently, there is no definitive, approved treatment for Long COVID. The current focus is on symptomatic care, using a multi-pronged approach. Notably, individualized rehabilitation is the mainstay of supportive care, rather than a universal cure. Treatment is initiated when symptoms appear, using a multi-pronged approach.
Management of individuals with Long COVID generally requires a multidisciplinary rehabilitation strategy. Care often involves collaboration among primary care physicians, physiotherapists, respiratory therapists, occupational therapists, and mental health professionals, among others, to address the diverse and persistent symptoms experienced by patients. For example, respiratory symptoms arising from persistent lung infection by SARS-CoV-2, require pulmonary rehabilitation, involving breathing retraining techniques, which help mitigate dyspnea. In case of cognitive impairment, cognitive behavioral therapy (CBT) has been found to be beneficial. Notably, mental problems like anxiety, depression, mood swings, psychosis, and post-traumatic stress disorder (PTSD) can be managed using established psychiatric or behavioral interventions coupled with pharmacological therapy.
PHARMACOLOGICAL INTERVENTIONS
Several pharmacological approaches have been explored for the management of Long COVID, targeting the potential mechanisms underlying persistent symptoms, which are presented below:
Antiviral drugs: Antiviral drugs are intended to eliminate residual viral particles or ongoing viral replication that may contribute to chronic inflammation in some patients. Commonly discussed agents include Paxlovid (a combination of nirmatrelavir and ritonavir), remdesivir, and molnupiravir. These medications interfere with the replication of SARS-CoV-2 by inhibiting key viral enzymes such as the viral protease or the RNA-dependent RNA polymerase (RdRp). Some clinical observations suggest that combination antiviral therapy, particularly the use of paxlovid together with remdesivir, may provide clinical improvement in certain individuals experiencing persistent post-COVID symptoms23.
Immunomodulators: Persistent immune activation and dysregulated cytokine signaling are considered possible contributors to Long COVID. Immunomodulatory therapies are therefore being investigated to restore immune balance. One example is low-dose naltrexone (LDN), an opioid receptor antagonist administered at much lower doses than those used for addiction treatment. At these doses, it is thought to exert immunomodulatory effects by reducing microglial activation and lowering the production of pro-inflammatory cytokines24.
Anti-inflammatory drugs: Medications that directly suppress inflammatory pathways may also help alleviate symptoms associated with chronic immune activation. Baricitinib, a Janus kinase (JAK1/JAK2) inhibitor, reduces inflammatory signaling by blocking the JAK-STAT pathway, thereby decreasing the expression of pro-inflammatory genes involved in cytokine-mediated responses. In addition, newer agents such as bezisterim are being investigated for their ability to selectively inhibit inflammatory processes while avoiding broad immunosuppressive effects. DFV890 is an oral small-molecule inhibitor of the NLRP3 inflammasome the blocks caspase-1 activation and targets exaggerated innate immune activation25. Dapansutrile (OLT1177) is another NLRP3 inhibitor that acts by direct interaction with its ATPase domain. Fluvoxamine is a selective serotonin reuptake inhibitor (SSRI) that modulates sigma-1 receptor and reduces cytokine production, thereby dampening inflammatory cascades.
Autoantibody neutralizing drugs: BC007 is an experimental agent that neutralizes G protein-coupled receptor (GPCR) autoantibodies, which are responsible for Long COVID autoimmunity. This drug reduces pathological antibody-driven signaling.
ANTIHISTAMINIC THERAPY
Antihistamine medications, including cetirizine and famotidine, help reduce histamine-mediated responses by limiting mast cell activation and subsequent histamine release. These agents are therefore often considered in the management of Mast Cell Activation Syndrome (MCAS), which has been reported as a common complication associated with Long COVID.
Gut-targeted therapies: Certain therapies aim to address increased intestinal permeability, commonly referred to as “leaky gut.” Larazotide is one such agent that acts by stabilizing the tight junctions of the intestinal epithelium, thereby helping to restore barrier integrity and reduce gut permeability. Early intervention may also help decrease the circulation of viral antigens and has been associated with symptomatic improvement in pediatric cases26.
Antioxidant therapies: Agents with antioxidant properties, such as L-arginine and vitamin C, have been investigated for their ability to counter oxidative stress and endothelial dysfunction observed in Long COVID. By supporting endothelial function, these compounds may contribute to improved vascular function and overall physiological balance27.
Cardiac and autonomic modulators: Several existing medications are being repurposed to address cardiovascular and autonomic symptoms seen in Long COVID. Ivabradine, an HCN channel inhibitor, is used in the treatment of POTS by lowering heart rate and improving tolerance to upright posture. Similarly, beta-blockers such as metoprolol may help manage cardiac manifestations and autonomic dysregulation associated with the condition28.
AI in long COVID management: Since Long COVID is a heterogeneous, relapsing-remitting disease, involving multiple organ systems and fluctuating symptoms, it complicates clinical recognition, risk stratification, follow-up intensity, and selection of supportive therapies and rehabilitation pathways.
AI encompasses machine learning (ML), natural language processing (NLP), and large language models (LLMs). These are increasingly being used to support Long COVID management in the undermentioned ways.
Case finding, risk stratification, and early triage: Early identification of individuals at risk for Long COVID enables timely follow-up and supportive interventions. ML models trained on large electronic health record (EHR) datasets have demonstrated moderate accuracy in predicting Long COVID based on demographic factors, comorbidities, acute-phase symptoms, and treatments. Predictive models help identifying clinically plausible risk factors such as age, obesity, chronic lung disease, and symptom burden. ML models can achieve useful discrimination while emphasizing the importance of interpretability and generalizability29. These tools are best positioned to aid triage and prioritization rather than serve as standalone diagnostic instruments.
Phenotyping and subtyping to guide personalized care pathways: Long COVID is increasingly recognized as a collection of overlapping syndromes rather than a single entity. ML-based clustering of electronic health record (HER) data has enabled the identification of reproducible subphenotypes reflecting dominant organ-system involvement. Four major post-acute subphenotypes have been identified, each associated with distinct clinical patterns and healthcare utilization30. Moreover, Long COVID subtypes derived using semantic similarity methods can be generalized across large US datasets. Temporal subphenotypes that capture symptom evolution over time have been identified using longitudinal data. Such AI-driven phenotyping can inform tailored diagnostic workups, referral pathways, and stratified clinical trials31.
Monitoring disease burden using wearables and digital biomarkers: Symptoms such as fatigue, dysautonomia, sleep disturbances, and post-exertional symptom exacerbation are difficult to quantify during intermittent clinic visits. Wearable devices provide continuous physiologic data, including heart rate, activity, and sleep patterns, which can be analyzed using ML to identify objective correlates of symptom burden. The feasibility of wearable-based monitoring in Long COVID is well established. However, there are challenges related to data quality and clinical validation32. Recent studies combining wearable data with patient-reported outcomes have shown improved detection of Long COVID compared with either modality alone33. These approaches may support remote monitoring, pacing strategies, and early identification of symptom exacerbations, provided that algorithms are rigorously validated.
AI enabled digital health platforms for self-management and clinician support: Long COVID management often relies on symptom tracking, behavioral strategies, rehabilitation, and longitudinal follow-up, making it well-suited to digital health solutions. Digital interventions have demonstrated potential benefits for physical and mental health management in post-COVID populations. Some platforms explicitly integrate AI to personalize feedback and support clinical decision-making. The SENSING-AI project exemplifies an AI-driven digital health solution designed to support adaptive self-management and clinician oversight through continuous patient-generated data34. While many such tools remain in early phases of evaluation, they illustrate scalable approaches to care coordination and monitoring.
Personalizing rehabilitation and recovery support: Rehabilitation is central to Long COVID care, particularly for functional limitation and deconditioning, while carefully avoiding symptom exacerbation in susceptible individuals. The AI can enhance rehabilitation programs by tailoring intensity, monitoring response, and identifying non-response or relapse. The ML-derived phenotypes can also help route patients to appropriate services, such as respiratory rehabilitation, autonomic clinics, or neurocognitive assessment, thereby optimizing resource utilization and patient outcomes35.
Operational decision support, documentation, and patient communication: The power of LLMs is being harnessed for clinical documentation, summarization of complex histories, and patient communication. In Long COVID, where multisystem symptoms and prolonged timelines are common, LLMs may assist clinicians by synthesizing records and drafting patient-facing instructions aligned with clinical decisions. However, evidence for direct clinical impact remains limited. Studies have highlighted both promise and ongoing concerns regarding accuracy, safety, and governance36. Consequently, the current best use lies in administrative and supportive roles rather than autonomous clinical decision-making.
Flip side of AI in Long COVID management: AI models for Long COVID are vulnerable to bias arising from underdiagnosis, variable coding practices, and inequitable access to care. Without safeguards, such models may exacerbate health disparities. Bias in medical AI is well documented, necessitating fairness auditing, transparent reporting, external validation, and human-in-the-loop oversight37. Privacy and data security are particularly critical when using wearables and patient-generated data.
COMPREHENSIVE LONG COVID CARE: THE JOHNS HOPKINS MODEL
Comprehensive care for Long COVID works best when it is structured, multidisciplinary, and rehabilitation-forward, because of the heterogeneous symptoms, spanning multiple organ systems.
At Johns Hopkins, including the Bayview campus ecosystem, the Post-Acute COVID-19 Team (PACT) was designed as an ambulatory framework that standardizes triage, evaluation, and longitudinal follow-up for three common survivor streams: (i) Post-ICU/critical illness, (ii) Post-hospitalization, and (iii) Non-hospitalized patients with persistent symptoms38. The clinic model focuses on the following aspects:
| • | Early phenotyping and risk stratification: These take into account the severity of acute illness, ICU exposure, pre-existing disease, and red-flag symptoms | |
| • | Stepwise diagnostics: These include cardiopulmonary testing, functional evaluation, and cognitive/mood screening, to avoid both under- and over-investigation | |
| • | Rehabilitation as a core therapy: This involves pacing/energy-management strategies and graded, individualized reconditioning rather than “one-size-fits-all” exercise prescriptions | |
| • | Tight referral loops: This ensures that patients do not bounce between siloed specialties |
The PACT operationalizes multidisciplinary rehabilitation by pairing pulmonary/critical care with physical medicine and rehabilitation at the center, then integrating services that commonly determine recovery trajectory. These include physiotherapy, occupational therapy, speech-language therapy, and if required, also cardiology, neurology, psychiatry/psychology, sleep medicine, and social work39. This team approach matters because Long-COVID disability is frequently a combined problem of impaired cardiopulmonary reserve, deconditioning, autonomic symptoms, cognitive-communication deficits, and psychological distress, requiring coordinated goals, shared outcome measures, and consistent patient education.
Thus, PACT functions as the program’s ‘care integrator’, as it provides a single point of access, ensures consistent triage and follow-up, aligns specialty input with rehabilitation plans, and reduces fragmented care. This approach is aligned with broader evidence mapping of post-COVID rehabilitation models internationally40.
Multidisciplinary post-COVID clinics are now common across North America and Europe, often built around internal medicine/pulmonary plus rehabilitation and mental health, reflecting the same care principles. In India, dedicated post-COVID services have emerged in major academic and corporate hospital systems. This need is underscored by Indian cohorts demonstrating persistent symptom burden after omicron-era infections41. Pragmatically, India’s scale and access constraints make pulmonary rehabilitation and tele-rehabilitation especially relevant, with Indian studies reporting improvements in function and quality-of-life measures following structured rehabilitation programs42.
CONCLUSION
Long COVID should be recognized as a complex, chronic condition requiring sustained clinical attention and health system adaptation. Continued investment in multidisciplinary care models, longitudinal research, and inclusive global data generation, especially from resource-limited settings, will be essential to mitigate the long-term impact of the pandemic and to improve outcomes for individuals living with the enduring consequences of COVID-19.
SIGNIFICANCE STATEMENT
Long COVID affects 10-30% of COVID-19 survivors, causing persistent, multi-system symptoms such as fatigue, cognitive impairment, and dyspnea, with higher risk in females, those with comorbidities, or severe acute illness. Its complex, multifactorial pathogenesis necessitates interdisciplinary, symptom-directed management. Models like Johns Hopkins PACT demonstrate the importance of coordinated care, rehabilitation, and data-driven strategies to improve outcomes and guide evidence-based treatment.
REFERENCES
- Tsampasian, V., H. Elghazaly, R. Chattopadhyay, M. Debski and T.K.P. Naing et al., 2023. Risk factors associated with post-COVID-19 condition: A systematic review and meta-analysis. JAMA Intern. Med., 183: 566-580.
- Parker, A.M., E. Brigham, B. Connolly, J. McPeake and A.V. Agranovich et al., 2021. Addressing the post-acute sequelae of SARS-CoV-2 infection: A multidisciplinary model of care. Lancet Respir. Med., 9: 1328-1341.
- Hanson, S.W., C. Abbafati, J.G. Aerts, Z. Al-Aly and C. Ashbaugh et al., 2022. Estimated global proportions of individuals with persistent fatigue, cognitive, and respiratory symptom clusters following symptomatic COVID-19 in 2020 and 2021. JAMA, 328: 1604-1615.
- Chen, C., S.R. Haupert, L. Zimmermann, X. Shi, L.G. Fritsche and B. Mukherjee, 2022. Global prevalence of post-coronavirus disease 2019 (COVID-19) condition or long COVID: A meta-analysis and systematic review. J. Infect. Dis., 226: 1593-1607.
- O'Mahoney, L.L., A. Routen, C. Gillies, W. Ekezie and A. Welford et al., 2023. The prevalence and long-term health effects of Long COVID among hospitalised and non-hospitalised populations: A systematic review and meta-analysis. eClinicalMedicine, 55.
- Grippo, F., G. Minelli, R. Crialesi, S. Marchetti, F. Pricci and G. Onder, 2024. Deaths related to post-COVID in Italy: A national study based on death certificates. Front. Med., 11.
- Arjun, M.C., A.K. Singh, D. Pal, K. Das and G. Alekhya et al., 2022. Characteristics and predictors of Long COVID among diagnosed cases of COVID-19. PLoS ONE, 17.
- Fernández-de-las-Peñas, C., J. Rodríguez-Jiménez, I. Cancela-Cilleruelo, A. Guerrero-Peral and J.D. Martín-Guerrero et al., 2022. Post-COVID-19 symptoms 2 years after SARS-CoV-2 infection among hospitalized vs nonhospitalized patients. JAMA Netw. Open, 5.
- Alkodaymi, M.S., O.A. Omrani, N.A. Fawzy, B. Abou Shaar and R. Almamlouk et al., 2022. Prevalence of post-acute COVID-19 syndrome symptoms at different follow-up periods: A systematic review and meta-analysis. Clin. Microbiol. Infect., 28: 657-666.
- Michelen, M., L. Manoharan, N. Elkheir, V. Cheng and A. Dagens et al., 2021. Characterising long COVID: A living systematic review. BMJ Global Health, 6.
- Xie, Y., E. Xu, B. Bowe and Z. Al-Aly, 2022. Long-term cardiovascular outcomes of COVID-19. Nat. Med., 28: 583-590.
- Lopez-Leon, S., T. Wegman-Ostrosky, C. Perelman, R. Sepulveda, P.A. Rebolledo, A. Cuapio and S. Villapol, 2021. More than 50 long-term effects of COVID-19: A systematic review and meta-analysis. Sci. Rep., 11.
- Carmona-Torre, F., A. Mínguez-Olaondo, A. López-Bravo, B. Tijero and V. Grozeva et al., 2022. Dysautonomia in COVID-19 patients: A narrative review on clinical course, diagnostic and therapeutic strategies. Front. Neurol., 13.
- Rahmati, M., R. Udeh, J. Kang, X. Dolja-Gore and M. McEvoy et al., 2025. Long-term sequelae of COVID-19: A systematic review and meta-analysis of symptoms 3 years post-SARS-CoV-2 infection. J. Med. Virol., 97.
- Ghafari, M., M. Hall, T. Golubchik, D. Ayoubkhani and T. House et al., 2024. Prevalence of persistent SARS-CoV-2 in a large community surveillance study. Nature, 626: 1094-1101.
- Su, Y., D. Yuan, D.G. Chen, R.H. Ng and K. Wang et al., 2022. Multiple early factors anticipate post-acute COVID-19 sequelae. Cell, 185: 881-895.
- Wu, X., M. Xiang, H. Jing, C. Wang, V.A. Novakovic and J. Shi, 2024. Damage to endothelial barriers and its contribution to long COVID. Angiogenesis, 27: 5-22.
- Dani, M., A. Dirksen, P. Taraborrelli, M. Torocastro, D. Panagopoulos, R. Sutton and P.B. Lim, 2021. Autonomic dysfunction in ‘long COVID’: Rationale, physiology and management strategies. Clin. Med., 21: e63-e67.
- Kenny, G., L. Townsend, S. Savinelli and P.W.G. Mallon, 2023. Long COVID: Clinical characteristics, proposed pathogenesis and potential therapeutic targets. Front. Mol. Biosci., 10.
- Wölfel, R., V.M. Corman, W. Guggemos, M. Seilmaier and S. Zange et al., 2020. Virological assessment of hospitalized patients with COVID-2019. Nature, 581: 465-469.
- Davis, H.E., G.S. Assaf, L. McCorkell, H. Wei and R.J. Low et al., 2021. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. eClinicalMedicine, 38.
- Zeraatkar, D., M. Ling, S. Kirsh, T. Jassal and M. Shahab et al., 2024. Interventions for the management of long COVID (post-COVID condition): Living systematic review. BMJ, 387.
- Bouhamdani, N., D. Bouhamdani, C. Léger, J. Stadler and N. Saulnier, 2025. Successful treatment of prolonged COVID-19 with remdesivir and nirmatrelvir/ritonavir in a patient with a history of diffuse large B-cell lymphoma: A case report. J. Egypt. Natl. Cancer Inst., 37.
- Tamariz, L., E. Bast, N. Klimas and A. Palacio, 2024. Low-dose naltrexone improves post-COVID-19 condition symptoms. Clin. Ther., 46: E101-E106.
- Gatlik, E., B. Mehes, E. Voltz, U. Sommer and E. Tritto et al., 2024. First-in-human safety, tolerability, and pharmacokinetic results of DFV890, an oral low-molecular-weight NLRP3 inhibitor. Clin. Transl. Sci., 17.
- Yonker, L.M., T. Gilboa, A.F. Ogata, Y. Senussi and R. Lazarovits et al., 2021. Multisystem inflammatory syndrome in children is driven by zonulin-dependent loss of gut mucosal barrier. J. Clin. Invest., 131.
- Izzo, R., V. Trimarco, P. Mone, T. Aloè and M.C. Marzani et al., 2022. Combining L-Arginine with vitamin C improves long-COVID symptoms: The LINCOLN survey. Pharmacol. Res., 183.
- Bonilla, H., M.J. Peluso, K. Rodgers, J.A. Aberg and T.F. Patterson et al., 2023. Therapeutic trials for long COVID-19: A call to action from the interventions taskforce of the RECOVER initiative. Front. Immunol., 14.
- Antony, B., H. Blau, E. Casiraghi, J.J. Loomba and T.J. Callahan et al., 2023. Predictive models of long COVID. eBioMedicine, 96.
- Zhang, H., C. Zang, Z. Xu, Y. Zhang and J. Xu et al., 2023. Data-driven identification of post-acute SARS-CoV-2 infection subphenotypes. Nat. Med., 29: 226-235.
- Dagliati, A., Z.H. Strasser, Z.S.H. Abad, J.G. Klann and K.B. Wagholikar et al., 2023. Characterization of long COVID temporal sub-phenotypes by distributed representation learning from electronic health record data: A cohort study. eClinicalMedicine, 64.
- Khondakar, K.R. and A. Kaushik, 2023. Role of wearable sensing technology to manage long COVID. Biosensors, 13.
- Uwakwe, C.K., E.S. Rangan, S. Kumar, G. Gutjahr and X. Miao et al., 2025. Longitudinal wearable sensor data enhance precision of Long COVID detection. PLOS Digital Health, 4.
- Fuster-Casanovas, A., L. Fernandez-Luque, F.J. Nuñez-Benjumea, A.M. Conde and L.G. Luque-Romero et al., 2022. An artificial intelligence-driven digital health solution to support clinical management of patients with Long COVID-19: Protocol for a prospective multicenter observational study. JMIR Res. Protoc., 11.
- He, J. and T. Yang, 2022. In the era of long COVID, can we seek new techniques for better rehabilitation? Chronic Dis. Transl. Med., 8: 149-153.
- Omar, M., G.N. Nadkarni, E. Klang and B.S. Glicksberg, 2024. Large language models in medicine: A review of current clinical trials across healthcare applications. PLOS Digital Health, 3.
- Cross, J.L., M.A. Choma and J.A. Onofrey, 2024. Bias in medical AI: Implications for clinical decision-making. PLOS Digital Health, 3.
- Brigham, E., J. O'Toole, S.Y. Kim, M. Friedman and L. Daly et al., 2021. The Johns Hopkins Post-Acute COVID-19 Team (PACT): A multidisciplinary, collaborative, ambulatory framework supporting COVID-19 survivors. Am. J. Med., 134: 462-467.e1.
- Santhosh, L., B. Block, S.Y. Kim, S. Raju and R.J. Shah et al., 2021. Rapid design and implementation of post-COVID-19 clinics. Chest, 160: 671-677.
- Décary, S., W. de Groote, C. Arienti, C. Kiekens and P. Boldrini et al., 2022. Scoping review of rehabilitation care models for post COVID-19 condition. Bull. World Health Organ., 100: 676-688.
- Arjun, M.C., A.K. Singh, P. Roy, M. Ravichandran and S. Mandal et al., 2023. Long COVID following Omicron wave in Eastern India—A retrospective cohort study. J. Med. Virol., 95.
- Bargaje, M.D., P. Sharma, J.D. Londhe, S.V. Patil and A.T. Anokar et al., 2024. Effectiveness of pulmonary rehabilitation in post-COVID-19 patients: A pre- and post-interventional study. Lung India, 41: 435-441.
How to Cite this paper?
APA-7 Style
Bharati,
K. (2026). Long COVID: Key Features and Current Trends in Management. Trends in Medical Research, 21(1), 23-39. https://doi.org/10.3923/tmr.2026.23.39
ACS Style
Bharati,
K. Long COVID: Key Features and Current Trends in Management. Trends Med. Res 2026, 21, 23-39. https://doi.org/10.3923/tmr.2026.23.39
AMA Style
Bharati
K. Long COVID: Key Features and Current Trends in Management. Trends in Medical Research. 2026; 21(1): 23-39. https://doi.org/10.3923/tmr.2026.23.39
Chicago/Turabian Style
Bharati, Kaushik.
2026. "Long COVID: Key Features and Current Trends in Management" Trends in Medical Research 21, no. 1: 23-39. https://doi.org/10.3923/tmr.2026.23.39

This work is licensed under a Creative Commons Attribution 4.0 International License.