Effect of Physalis angulata L. Leaves on Free Radicals: Antioxidant Study
| Received 17 Oct, 2023 |
Accepted 30 Nov, 2023 |
Published 02 Dec, 2023 |
Background and Objective: Physalis angulata is used in ethnomedicine for the treatment of numerous diseases. The free radical scavenging effect of Physalis angulataL. leaves ethanolic extract and extract fractions (n-butanol, ethyl acetate and n-hexane fractions) was determined in this study. Materials and Methods: Free radicals and antioxidant properties of Physalis angulataL. leaves ethanolic extract and extract fractions were investigated using in vitromodel. Total phenol, flavonoid, total antioxidant capacity, 2,2-Diphenyl-1-Picrylhydrazyl (DPPH), nitric oxide and 2,2-Azino-Bis-3-Ethylbenzothiazoline-6-Sulphonic Acid (ABTS) assays were carried out using the spectrophotometric method. One-Way Analysis of Variance (ANOVA) and Dunnett post hoc tests at 95% level of significance (p<0.05) and n = 5 (sample size) were used for statistical analysis. Results: The Physalis angulataleaves ethanolic extract and extract fractions exerted free radical scavenging properties. The ethyl acetate fraction showed a higher (significant at p<0.05) total antioxidant, (55.71 mg ascorbic acid equivalent/g of extract), flavonoid (37.88 mg quercetin equivalent/g of extract) and phenol (48.14 mg gallic acid equivalent/g of extract) content when compared with the other extracts. Ethyl acetate extract fraction also had the highest (significant at p<0.05), DPPH (77.87%), ABTS (56.63%) and nitric oxide (56.28%) free radicals scavenging effect when compared with the other plant extracts. Conclusion: The Physalis angulataleaves ethanolic extract and extract fractions exerted free radicals scavenging activities and it could be explored as a medicine for the treatment of diseases caused by free radicals.
| Copyright © 2023 Sunday and Adeyemi. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Plants with medicinal properties have been used since ancient times for the treatment of numerous diseases due to the presence of antioxidants that help in scavenging free radicals1. Plants contain compounds known as antioxidants that scavenge free radicals which might cause damage to body cells2,3. Oxidative reactions in the body lead to the production of free radicals which helps in metabolic reactions taking place in body cells4. Free radicals in high concentration in the body can cause damage to cells and these might lead to diseases such as cancer, diabetes, quick aging and nervous system disorder5-8. Studies carried out recently on antioxidants obtained from different parts of plants reported that plants may be used for the treatment of diseases caused by free radicals9,10.
Physalis angulata L. belonging to the family Solanaceae is a plant used in ethno-medicine for the treatment of numerous diseases due to its medicinal properties. The plant is found in tropical, subtropical and temperate regions, these include Nigeria, India, Brazil, South America and Central America. In English language, P. angulata is known as Wild gooseberry.
Physalis angulata is called Saadi-birii and Koropo in Hausa and Yoruba languages respectively. In ethnomedicine, different parts of P. angulata are used for the treatment of disease11,12 in numerous countries including Nigeria13, Kenya14, Bolivia15, Indonesia16, India17, Equatorial Guinea18 and Brazil19. Physalis angulata is used for the treatment of fever, malaria, cough, acne, asthma, diabetes11, rashes, gonorrhea, stomach ache, hepatitis, anaemia and for pain relief.
There are reports from studies that Physalis angulata has antimicrobial11,20, anticancer21, immunomodulatory22, anti-inflammatory23, neuroprotective24, anti-oxidant25 and anti-malarial activity26.
In this study, the free radical scavenging properties of Physalis angulata leaves ethanolic extract and extract fractions (n-butanol, ethyl acetate and n-hexane fractions) were evaluated by determining the total antioxidant contents (flavonoid, phenol and total antioxidant capacity) and the percentage inhibition of free radicals (2,2-diphenyl-1-picrylhydrazyl, nitric oxide and 2,2-azino-bis-3-ethylbenzothiazoline-6-sulphonic acid) using spectrophotometric technique.
MATERIALS AND METHODS
Study area and duration: This study was carried out from June, 2022 to May, 2023 at Department of Pharmacology, Faculty of Pharmacy, Obafemi Awolowo University, Ile-Ife, Osun State, Nigeria and Medical Biotechnology Department, National Biotechnology Development Agency, Abuja, Nigeria.
Plant material: Physalis angulata leaves were obtained from Ogbomoso, Oyo State, Nigeria. The plant was authenticated at Ife Herbarium, Department of Botany, Obafemi Awolowo University (O.A.U), Ile-Ife, Osun State, Nigeria.
Ethanolic extraction: Physalis angulata leaves were washed under clean running tap water and then oven-dried (40oC). The dried plant sample (2 kg) was ground into powder, soaked in ethanol (70%) for 72 hrs and filtered using Whatman No.1 filter paper. The filtrate was concentrated into a solid paste using a rotary evaporator (Rotavapor R-100, BUCHI, Switzerland) and thereafter air-dried before proceeding with the experiment. The dried plant extract was used for the study27.
Fractionation: Physalis angulata ethanolic extract was fractionated using n-hexane, followed by ethylacetate and lastly n-butanol28. Thereafter, the ethanolic extract fractions were each air-dried before using it for the experiment.
Assays for antioxidants: As 1.0 mg mL–1 of the ethanolic extract, n-hexane fraction, ethylacetate fraction and n-butanol fraction of P. angulata were used for total antioxidant capacity, flavonoid content and phenol content assays.
Total antioxidant capacity assay: A volume of 1.0 mL of reagent mixture (0.6 M sulphuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate) was added to 1.0 mL of the samples in different test tubes and then incubated in a water bath at 95°C for 90 min. Thereafter, the mixture was cooled to room temperature and the absorbance was measured at 695 nm against a reagent blank10. The total antioxidant capacity of the extracts expressed as mg standard (ascorbic acid) equivalent per gram of the plant extract was calculated using the following equation Eq. 1:
| (1) |
Where:
| T | = | Total antioxidant in mg standard | |
| c | = | Concentration of standard established from the calibration curve in mg mL–1 | |
| v | = | Volume of the extract in mL | |
| m | = | Weight of the extract in gm |
Total flavonoid content assay: The 5% sodium nitrite (0.3 mL) was added to a volume of 0.5 mL of the samples in different test tubes and it was left for 5 min before adding 0.3 mL of aluminium chloride (10%). As 1 M sodium hydroxide (2.0 mL) was thereafter added to the mixture after six min. Absorbance was read at 510 nm and Eq. 1 was used to calculate the total flavonoid contents as mg quercetin (standard) equivalents/gram of extract10. In Eq. 1, T = Total flavonoids content in mg quercetin.
Total phenol content assay: As 1 M Folin-Ciocalteu’s phenol reagent (8.0 mL) was added to 0.2 mL of each plant extract in different test tubes. Saturated sodium carbonate solution (8% w/v in distilled water) at a volume of 1.0 mL was thereafter added to the mixture and it was kept in the dark for 30 min. Absorbance was read at 765 nm and Eq. 1 was used to calculate the total phenol content as mg gallic acid (standard) equivalent/gram of extract29. In Eq. 1 and T = Total phenol content in mg gallic acid (standard).
Assays for scavenging free radicals: The 2,2-diphenyl-1-picrylhydrazyl, nitric oxide and 2,2-azino-bis-3-ethylbenzothiazoline-6-sulphonic acid assays were carried out using 62.5, 125, 250, 500 and 1000 μg ethanolic extract/mL and 62.5 μg of each extract fraction/mL.
Assay for 2, 2-Diphenyl-1-Picrylhydrazyl (DPPH): The 0.3 mM DPPH in methanol (1.0 mL) was added to 1.0 mL of each plant extract. The solution was mixed and incubated for 30 min in the dark. Absorbance was read at 517 and Eq. 2 was used to calculate the radical scavenging activity in percentage (RSA%)9.
| (2) |
Assay for nitric oxide: The 3.0 mL of sodium nitroprusside (10 mm) in phosphate-buffered saline (pH 7.4) was added to 1.0 mL of each plant extract in a test tube and then incubated at 25oC for 1 hr. Thereafter Griess reagent (5.0 mL) was added to the mixture. Absorbance was read at 546 nm and Eq. 2 was used to calculate the radical scavenging activity in percentage (RSA%)30,31.
2, 2-azino-bis-3-ethylbenzothiazoline6-sulphonic Acid (ABTS) assay: The ABTS (7 mM) was mixed with 7 mM potassium per sulphate in a ratio 2:1 and the mixture was allowed to stand in the dark at room temperature for 16 hrs. The 70% ethanol was then added to the mixture before using the ABTS working solution. The ABTS radical scavenging analysis was performed by mixing 2.0 mL of the ABTS working solution with 20 μL of the different concentrations (62.5, 125, 250, 500 and 1000 μg mL–1 of methanol) of plant extracts/standard solution (trolox). The negative control was prepared by adding 20 μL of 70% ethanol in 2.0 mL of ABTS. After 1 min the absorbance was measured spectrophotometrically at 734 nm32. The readings obtained were used to calculate the percentage of radical scavenging activity (RSA) as follows:
Statistical analysis: The results were analyzed using One-Way Analysis of Variance (ANOVA) followed by Dunnett post hoc multiple comparisons tests at 95% (p<0.05) level of significance using Primer (version 3.01). All results were expressed as Mean±Standard Error of the Mean (SEM). The 50% inhibition (IC50) of the plant extract was calculated from a graph plotted using Excel.
RESULTS
Antioxidant activity of Physalis angulata leaves: The results of the study show that a moderate amount of flavonoid and phenol are present in Physalis angulata leaves ethanolic extract and extract fractions (n-butanol, ethyl acetate and n-hexane extract fractions). The ethyl acetate extract fraction showed a significant (p<0.05) increase in the total flavonoid and phenol content when compared with the ethanolic extract and other extract fractions (Table 1).
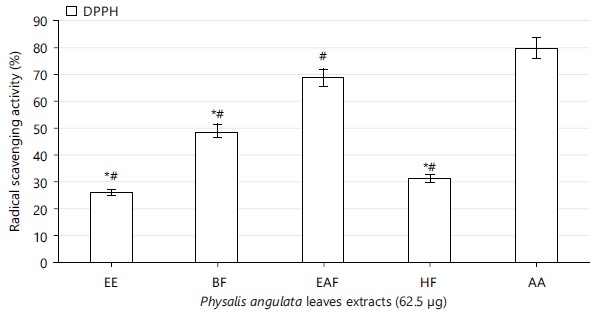
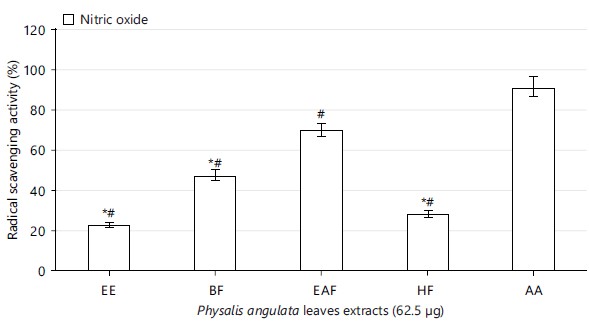
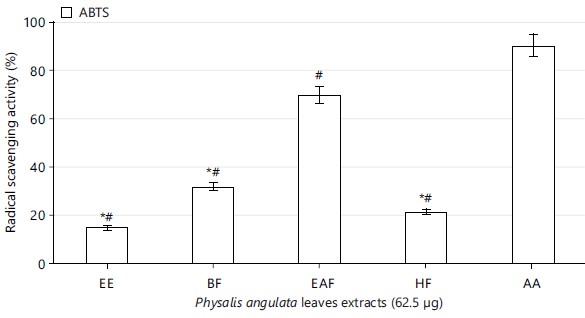
Radical scavenging effect of Physalis angulata leaves: The concentrations of Physalis angulata leaves ethanolic extract used for this study showed free radical scavenging property with increasing concentrations, significant at p<0.05 (Table 2). The extract fractions (n-hexane, ethyl acetate and n-butanol) also scavenged the free radicals (Fig. 1-3). The ethyl acetate extract fraction scavenged the free radicals more than the ethanolic extract and other fractions of the extract, significant at p<0.05 (Fig. 1-3).
|
| Table 1: | Antioxidant activity of Physalis angulata leaves extracts | |||
| Ethanolic extract and extract fractions (62.5 μg) | TAC (mg AAE/g) |
TF (mg QE/g) |
TP (mg GAE/g) |
| Ethanolic extract | 24.92±0.22* |
16.11±0.21* |
20.64±0.23* |
| n-Butanol fraction | 31.14±0.16* |
20.05±0.20* |
28.75±0.19* |
| Ethylacetate fraction | 55.71±0.19 |
37.88±0.17 |
48.14±0.20 |
| n-Hexane fraction | 22.20±0.20* |
18.06±0.24* |
16.90±0.21* |
| Mean±SEM, n = 3, TAC: Total antioxidant capacity, TF: Total flavonoid, TP: Total phenol, mg AAE/g: Mg ascorbic acid equivalent/g of extract, mg QE/g: Mg quercetin equivalent/g of extract, mg GAE/g: Mg gallic acid equivalent/g of extract and *Significantly different from ethyl acetate extract fraction at p<0.05 | |||
|
|
| Table 2: | Radical scavenging activity of Physalis angulata leaves ethanolic extract | |||
| Extract concentration (mg) | DPPH |
ABTS |
Nitric oxide |
| 62.5 μg | 26.11±0.20* |
14.57±0.23* |
23.12±0.14* |
| 125 μg | 30.64±0.14* |
17.24±0.25* |
29.08±0.58* |
| 250 μg | 36.32±0.16* |
26.78±0.20* |
36.90±0.53* |
| 500 μg | 44.10±0.18* |
35.19±0.21* |
41.71±0.54* |
| 1000 μg | 50.83±0.14* |
40.10±0.20* |
48.19±0.58* |
| Ascorbic acid (100 μg) | 79.94±0.31 |
88.92±0.41 |
92.64±0.21 |
| Mean±SEM, n = 3, DPPH: 2, 2-Diphenyl-1-Picrylhydrazyl, ABTS: 2,2-Azino-Bis-3-Ethylbenzothiazoline-6-Sulphonic acid and *Significantly from Ascorbic acid (100 μg) at p<0.05 | |||
DISCUSSION
Physalis angulata leaves ethanolic extract and extract fractions showed the presence of antioxidants such as flavonoid and phenol (Table 1). The plant extracts also exerted a potent free radical scavenging effect.
Physalis angulata leaves ethanolic extract and fractions scavenged free radicals present in 2,2-diphenyl-1-picrylhydrazyl, nitric oxide and 2,2-azino-bis-3-ethylbenzothiazoline-6-sulphonic acid (Table 2, Fig. 1 to 3). The results on antioxidant properties of Physalis angulata leaves gotten from this study was similar to reports from previous studies carried out on different species of the genus Physalis. It was reported that Physalis patula, Physalis solanacea, Physalis hederifolia and Physalis subulata exhibited antioxidant activities and phenolic compounds were also present in the plant33.
It was also reported from previous studies that Physalis angulata ripe fruits showed total phenolic content and antioxidant activity values that are higher than that of Physalis peruviana ripe fruit34. Antioxidant studies carried out on the methanolic extract of Physalis angulata stem, root, leaves and stem, reported that the plant exerted antioxidant activities35. Also, the leaves and fruits of the methanolic extract were reported to have a higher flavonoid and phenolic content35.
Phenolic compounds are secondary metabolites with antioxidant activities found in plants. Antioxidants function as protective substances against diseases caused by excess free radicals in the body2,10. In this study, the ethyl acetate extract fraction of Physalis angulata leaves exerted a significantly higher flavonoid content, phenol content, total antioxidant capacity and free radical scavenging effect when compared with the ethanolic extract and other extract fractions (n-butanol fraction and n-hexane fraction). Physalis angulata has shown several biological activities that may be due to the presence of antioxidants.
Previous studies carried out on Physalis angulata reported that the fruits have immunomodulation, antioxidant and anti-inflammation properties36,37. The leaves are reported to have an antidiabetic effect38. The ethanolic extract of Physalis angulata fruits is reported to have anti-hypertension and anti-hyperglycemic effects39.
The results from this study showed that Physalis angulata leaves ethanolic extract and extract fractions exerted potent free radicals scavenging effect and the ethyl acetate extract fraction had the highest activity. These results suggest that the plant extract may be used for the treatment of diseases such as cardiovascular disease, diabetes and cancer caused by free radicals. The free radicals scavenging effect and antioxidant properties of Physalis angulata leaves were determined in this study. Further studies can be carried out in order to isolate the active compound responsible for the free radicals scavenging effect of Physalis angulata leaves.
CONCLUSION
This study shows that Physalis angulata leaves ethanolic extract and extract fractions exerted a free radical scavenging effect due to its antioxidant properties. The deductions from this study suggest that Physalis angulata leaves can be used for the treatment of diseases that are caused by free radicals which include cardiovascular disease and cancer. The authors recommend that further studies be carried out to isolate the active compound(s) responsible for the free radical scavenging effect of P. angulata leaves and these might provide a better therapy for diseases caused by free radicals.
SIGNIFICANCE STATEMENT
This study discovered that the ethyl acetate extract fraction of Physalis angulata leaves exerted a higher free radical scavenging effect, total antioxidant capacity, total flavonoid and total phenol content when compared with the ethanolic extract and other extract fractions (n-butanol fraction and n-hexane fraction). This study will help researchers carry out activity-guided research on the leaves of Physalis angulata using various solvent extracts that may lead to the development of new therapies that can be used for the treatment of chronic oxidative stress-related diseases.
REFERENCES
- Sabli, F., M. Mohamed, A. Rahmat, H. Ibrahim and M.F. Abu-Bakar, 2012. Antioxidant properties of selected Etlingera and Zingiber species (Zingiberaceae) from Borneo Island. Int. J. Biol. Chem., 6: 1-9.
- Yamagishi, S.I. and T. Matsui, 2011. Nitric oxide, a janus-faced therapeutic target for diabetic microangiopathy-friend or foe? Pharmacol. Res., 64: 187-194.
- Wu, Y.Y., W. Li, Y. Xu, E.H. Jin and Y.Y. Tu, 2011. Evaluation of the antioxidant effects of four main theaflavin derivatives through chemiluminescence and DNA damage analyses. J. Zhejiang Univ. Sci. B, 12: 744-751.
- Valko, M., D. Leibfritz, J. Moncol, M.T.D. Cronin, M. Mazur and J. Telser, 2007. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol., 39: 44-84.
- Valko, M., M. Izakovic, M. Mazur, C.J. Rhodes and J. Telser, 2004. Role of oxygen radicals in DNA damage and cancer incidence. Mol. Cell. Biochem., 266: 37-56.
- Kinnula, V.L. and J.D. Crapo, 2004. Superoxide dismutases in malignant cells and human tumors. Free Radical Biol. Med., 36: 718-744.
- Hyun, D.H., J.O. Hernandez, M.P. Mattson and R. de Cabo, 2006. The plasma membrane redox system in aging. Ageing Res. Rev., 5: 209-220.
- Sas, K., H. Robotka, J. Toldi and L. Vécsei, 2007. Mitochondria, metabolic disturbances, oxidative stress and the kynurenine system, with focus on neurodegenerative disorders. J. Neurol. Sci., 257: 221-239.
- Huang, B., H. Ke, J. He, X. Ban, H. Zeng and Y. Wang, 2011. Extracts of Halenia elliptica exhibit antioxidant properties in vitro and in vivo. Food Chem. Toxicol., 49: 185-190.
- Sasikumar, V. and P. Kalaisezhiyen, 2014. Evaluation of free radical scavenging activity of various leaf extracts from Kedrostis foetidissima (Jacq.) Cogn. Biochem. Anal. Biochem., 3.
- Osho, A., T. Adetunji, S.O. Fayemi and D.P. Moronkola, 2010. Antimicrobial activity of essential oils of Physalis angulata L. Afr. J. Tradit. Complementary Altern. Med., 7: 303-306.
- de Rosso, V.V. and A.Z. Mercadante, 2007. Identification and quantification of carotenoids, By HPLC-PDA-MS/MS, from amazonian fruits. J. Agric. Food Chem., 55: 5062-5072.
- Lawal, I.O., N.E. Uzokwe, A.B.I. Igboanugo, A.F. Adio and E.A. Awosan et al., 2010. Ethno medicinal information on collation and identification of some medicinal plants in Research Institutes of South-West Nigeria. Afr. J. Pharm. Pharmacol., 4: 1-7.
- Geissler, P.W., S.A. Harris, R.J. Prince, A. Olsen and R.A. Odhiambo et al., 2002. Medicinal plants used by Luo mothers and children in Bondo District, Kenya. J. Ethnopharmacol., 83: 39-54.
- Bourdy, G., S.J. DeWalt, L.R. Chávez de Michel, A. Roca and E. Deharo et al., 2000. Medicinal plants uses of the Tacana, an Amazonian Bolivian ethnic group. J. Ethnopharmacol., 70: 87-109.
- Roosita, K., C.M. Kusharto, M. Sekiyama, Y. Fachrurozi and R. Ohtsuka, 2008. Medicinal plants used by the villagers of a Sundanese community in West Java, Indonesia. J. Ethnopharmacol., 115: 72-81.
- Reddy, C.S., K.N. Reddy, E.N. Murthy and V.S. Raju, 2009. Traditional medicinal plants in Seshachalam Hills, Andhra Pradesh, India. J. Med. Plants Res., 3: 408-412.
- Akendengue, B., 1992. Medicinal plants used by the Fang traditional healers in Equatorial Guinea. J. Ethnopharmacol., 37: 165-173.
- Agra, M.F., G.S. Baracho, K. Nurit, I.J.L.D. Basílio and V.P.M. Coelho, 2007. Medicinal and poisonous diversity of the flora of “Cariri Paraibano”, Brazil. J. Ethnopharmacol., 111: 383-395.
- Silva, M.T.G., S.M. Simas, T.G.F.M. Batista, P. Cardarelli and T.C.B. Tomassini, 2005. Studies on antimicrobial activity, in vitro, of Physalis angulata L. (Solanaceae) fraction and physalin B bringing out the importance of assay determination. Mem. Inst. Oswaldo Cruz, 100: 779-782.
- Hwang, J.K., J.S. Shim and J.Y. Chung, 2004. Anticariogenic activity of some tropical medicinal plants against Streptococcus mutans. Fitoterapia, 75: 596-598.
- Soares, M.B.P., D. Brustolim, L.A. Santos, M.C. Bellintani and F.P. Paiva et al., 2006. Physalins B, F and G, seco-steroids purified from Physalis angulata L., inhibit lymphocyte function and allogeneic transplant rejection. Int. Immunopharmacol., 6: 408-414.
- Vieira, A.T., V. Pinho, L.B. Lepsch, C. Scavone and I.M. Ribeiro et al., 2005. Mechanisms of the anti-inflammatory effects of the natural secosteroids physalins in a model of intestinal ischaemia and reperfusion injury. Br. J. Pharmacol., 146: 244-251.
- Kuboyama, T., C. Tohda and K. Komatsu, 2005. Neuritic regeneration and synaptic reconstruction induced by withanolide A. Br. J. Pharmacol., 144: 961-971.
- Choi, E.M. and J.K. Hwang, 2005. Effect of some medicinal plants on plasma antioxidant system and lipid levels in rats. Phytother. Res., 19: 382-386.
- Abe, F., S. Nagafuji, M. Okawa and J. Kinjo, 2006. Trypanocidal constituents in plants 6. Minor withanolides from the aerial parts of Physalis angulata. Chem. Pharm. Bull., 54: 1226-1228.
- Sunday, R.M., O.R. Ilesanmi and E.M. Obuotor, 2016. Acute and sub-chronic oral toxicity of Anthocleista vogelii (cabbage tree) root hydroethanolic extract in albino rats. Br. J. Pharm. Res., 12.
- Jahan, N., M.S. Parvin, N. Das, M.S. Islam and M.E. Islam, 2014. Studies on the antioxidant activity of ethanol extract and its fractions from Pterygota alata leaves. J. Acute Med., 4: 103-108.
- Sunday, R.M., E.M. Obuotor and A. Kumar, 2019. Antioxidant activities of Asparagus adscendens root ethanolic extract and fractions using in vitro models. Trends Appl. Sci. Res., 14: 199-204.
- Green, L.C., D.A. Wagner, J. Glogowski, P.L. Skipper, J.S. Wishnok and S.R. Tannenbaum, 1982. Analysis of nitrate, nitrite and [15N] nitrate in biological fluids. Anal. Biochem., 126: 131-138.
- Boora, F., E. Chirisa and S. Mukanganyama, 2014. Evaluation of nitrite radical scavenging properties of selected Zimbabwean plant extracts and their phytoconstituents. J. Food Process., 2014.
- Re, R., N. Pellegrini, A. Proteggente, A. Pannala, M. Yang and C. Rice-Evans, 1999. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biol. Med., 26: 1231-1237.
- Medina-Medrano, J.R., N. Almaraz-Abarca, M.S. González-Elizondo, J.N. Uribe-Soto, L.S. González-Valdez and Y. Herrera-Arrieta, 2015. Phenolic constituents and antioxidant properties of five wild species of Physalis (Solanaceae). Bot. Stud., 56.
- de Oliveira, A.M., L.N. Malunga, C.A. Perussello, T. Beta and R.H. Ribani, 2020. Phenolic acids from fruits of Physalis angulata L. in two stages of maturation. South Afr. J. Bot., 131: 448-453.
- Krishna, T.M., R. Vadluri and E.M. Kumar, 2013. In vitro determination of antioxidant activity of Physalis angulata Lnn. Int. J. Pharma Biol. Sci., 4: 541-549.
- Adnyana, I.K., E. Yulinah, N. Maeistuti and F. Setiawan, 2014. Evaluation of ethanolic extracts of mullaca (Physalis angulata L.) herbs for treatment of lupus disease in mice induced pristane. Procedia Chem., 13: 186-193.
- Kusumaningtyas, R., N. Laily and P. Limandha, 2015. Potential of ciplukan (Physalis angulata L.) as source of functional ingredient. Procedia Chem., 14: 367-372.
- Kasali, F.M., C.N. Fokunang, J. Ngoupayo, E. Tembe-Fokunang and B. Ngameni et al., 2016. Evaluation of the antidiabetic properties of hydro-alcoholic extract and its fractions from Physalis peruviana L. leaves on streptozotocin-induced diabetic Wistar rats. J. Dis. Med. Plants, 2: 67-73.
- da Silva Pinto, M., L.G. Ranilla, E. Apostolidis, F.M. Lajolo, M.I. Genovese and K. Shetty, 2009. Evaluation of antihyperglycemia and antihypertension potential of native peruvian fruits using in vitro models. J. Med. Food, 12: 278-291.
How to Cite this paper?
APA-7 Style
Sunday,
R.M., Adeyemi,
O.I. (2023). Effect of Physalis angulata L. Leaves on Free Radicals: Antioxidant Study. Trends in Medical Research, 18(1), 179-186. https://doi.org/10.3923/tmr.2023.179.186
ACS Style
Sunday,
R.M.; Adeyemi,
O.I. Effect of Physalis angulata L. Leaves on Free Radicals: Antioxidant Study. Trends Med. Res 2023, 18, 179-186. https://doi.org/10.3923/tmr.2023.179.186
AMA Style
Sunday
RM, Adeyemi
OI. Effect of Physalis angulata L. Leaves on Free Radicals: Antioxidant Study. Trends in Medical Research. 2023; 18(1): 179-186. https://doi.org/10.3923/tmr.2023.179.186
Chicago/Turabian Style
Sunday, Rita, Maneju, and Oluwole Isaac Adeyemi.
2023. "Effect of Physalis angulata L. Leaves on Free Radicals: Antioxidant Study" Trends in Medical Research 18, no. 1: 179-186. https://doi.org/10.3923/tmr.2023.179.186

This work is licensed under a Creative Commons Attribution 4.0 International License.