Prevalence, Clinical Profile and Determinants of COVID-19 Vaccination and SARS-CoV-2 Breakthrough Infection in Douala, Cameroon
| Received 20 Aug, 2023 |
Accepted 14 Sep, 2023 |
Published 23 Oct, 2023 |
Background and Objective: Vaccination is the most cost-effective public health intervention to control diseases such as the current COVID-19 pandemic. This study aimed to determine the prevalence, clinical profile and determinants of COVID-19 vaccination uptake and SARS-CoV-2 breakthrough infection. Materials and Methods: A cross-sectional study was conducted in Douala, Cameroon from January, to September, 2022. A pretested structured questionnaire was used to collect sociodemographic, anthropometric and clinical data of participants during individual interviews. Additionally, findings on COVID-19 vaccination and RT-qPCR based SARS-CoV-2 molecular testing of fully COVID-19 vaccinated patients from medical records. Statistical analyses were performed using StatView, SPSS and GraphPad software. The level of statistical significance was set at p<0.05. Results: The overall prevalence of COVID-19 vaccination uptake was 18.6%. The prevalence of cough (p = 0.01) and sore throat (p = 0.03) was higher in unvaccinated patients. The chances of getting vaccinated were increased in those with higher education, elderly people and those with a history of COVID-19 infection. Vaccine breakthrough infections were found at a prevalence of 5.41% and were mainly seen in elderly patients with comorbidities (obesity, hypertension). Conclusion: There is a need for adequately tailored information campaigns to sensitize Cameroonian populations on the positive impact of COVID-19 vaccination, especially those with low levels of education.
| Copyright © 2023 Moguem Soubgui et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
The emergence of pathogens in humans is greatly due to an increased human-animal contact through factors such as expansion of human populations to wildlife and their environment1. It is likely through this way that a large number of life-threatening pathogens have emerged in humans. The last example is the emergence of the Severe Acute Respiratory Syndrome virus Syndrome Coronavirus (SARS-CoV-2), a betacoronavirus responsible for the current Coronavirus Disease 2019 pandemic (COVID-19)2,3. The current global statistics indicate that an estimated 630 million confirmed SARS-CoV-2 cases and ~6.5 million deaths all over the world, as of November 1st, 20222 Clinical presentation of COVID-19 is diverse with clinical spectrum ranging from asymptomatic carriage of the SARS-CoV-2 to severe forms and deaths4,5.
Control measures have been proposed and developed to curb COVID-19 and these include mainly behavioural change-related attitudes such as wearing mask and handwashing6,7. To date, there are no efficient therapeutics against COVID-19, even though a myriad of options has been evaluated so far (e.g., repurposed drugs, monoclonal antibodies)4. Vaccination remains the most effective public health intervention to control infectious diseases. A modelling study pointed out that COVID-19 vaccination saved tens of millions of lives globally8. A limited number of COVID-19 vaccines are currently available and these are either endorsed by the World Health Organisation (WHO), food drug administration and some countries (e.g., China, Russia). The main COVID-19 vaccines include the Oxford/AstraZeneca (ChAdOx1-S [recombinant]) vaccine, the Janssen Ad26.COV2.S COVID-19 vaccine, The moderna COVID-19 (mRNA-1273) vaccine and The Pfizer BioNTech (BNT162b2) COVID-19 vaccine9-11.
Existing evidences suggest that current COVID-19 vaccines are safe and efficient to prevent complications and deaths in COVID-19 patients12-14. Even though not avoiding SARS-CoV-2 transmission to humans, COVID-19 vaccines are moderately-to-highly effective against the main Alpha, Beta and Delta lineages of the SARS-CoV-212-14. Again, side effects reported in COVID-19 vaccinated people are generally mild (e.g, fever, fatigue, pain)9-11. Unfortunately, providing and coverage rates with COVID-19 vaccines are unjustly unequal between developed and developing countries, with dramatically low rates in resource-constrained areas such as African countries. Furthermore, a systematic review and meta-analysis outlined relatively low global acceptance and uptake of COVID-19 vaccination, with values of 67.8 and 42.3%, respectively15. The COVID-19 is still an important public health concern in Cameroon, despite the arsenal of strategies deployed to control the pandemic16,17. A national plan has been defined to implement and scale up COVID-19 vaccination in the country through the COVID-19 Vaccines Global Access (COVAX) Facility initiative, in association with, WHO18.
The present study was designed to determine coverage level, clinical profile and determinants of COVID-19 vaccination in Douala, Cameroon. Additionally, the prevalence and characteristics of SARS-CoV-2 breakthrough infections were also determined.
MATERIALS AND METHODS
Study design and sites: A hospital-based cross sectional study was conducted in the town of Douala, Littoral Region, Cameroon from January to September, 2022. Douala is the main business and most populous town of the country, which is characterized by a high ethnic diversity mainly represented by Duala, Bassa and Bamileke groups19. In the early of COVID-19 pandemics, the Government of Cameroon encouraged the implementation of control measures including vaccination. Sites of seven health facilities were selected in this study to recruit participants namely Bangue District Hospital, Boko Health Care Centre, Bonassama District Hospital, Cité des Palmiers District Hospital, Deido District Hospital, New Bell District Hospital and Nylon District Hospital. The selection of health facilities as study sites was driven by high attendance rates in these facilities and the implementation of diagnostic and treatment plateforms for COVID-19 management.
Participants and sample size: All Cameroonian patients coming for routine check-up or attending outpatient departments of either of the above mentioned health facilities, of both sexes, aged more than 18 years old, living permanentlyin Douala and who gave their consent to participate in the study. In contrast, individuals who declined the invitation to take part in the study, reluctant to sign written informed consent forms and foreigners were not enrolled. In order to prevent the risk of selection or information biases, the selection of participants was based on a consecutive inclusion using random sampling approach. The minimum sample size reauired for the study was computed using the Lwanga and Lemeshow’s formula20. The statistical parameters included level of confidence (95%), a 10% non-response rate and proportion of COVID-19 vaccination coverage (42.3%)15. The minimum sample size for the study was estimated at n = 412.5 ≈413 participants.
Data collection and management: An investigator-administered pretested structured questionnaire was used to collect data of interest during 20 min individual interviews. Data were collected by master and final-year doctoral students. The first part of the questionnaire was designed to collect sociodemographic information (gender, age, occupation, marital status and level of education). The second part captured clinical and anthropometric information including comorbidities (i.e., the co-occurrence of more than health disorder in the same patient). The third part focused on COVID-19 related information such as history of SARS-CoV-2 infection, COVID-19 vaccination, nature and number of doses of the COVID-19 vaccine.
Clinical signs and symptoms presented by participants were diagnosed and reported by medical doctors. Anthropometric data were weight, height and body mass index (BMI). The Quetelet’s formula was used to calculate BMI (kg m–2) = weight/(height)2. Obesity was defined as BMI >25 kg m–2. A patient was considered fully COVID-19 vaccinated if he/she had received the number of doses required for a given vaccine. For instance, the required dosage for the Oxford/AstraZeneca COVID-19 vaccine is two doses given intramuscularly (0.5 mL each) with an interval of 8-12 weeks7,18.
Sources of information included patients medical records, vaccination card, declaration of patients and discussion with medical doctors. Finally, findings on RT-qPCR based SARS-CoV-2 molecular testing of fully COVID-19 vaccinated patients were collected from medical records. This information allowed to determine rate of SARS-CoV-2 breakthrough infection (i.e., presence of SARS-CoV-2 infection among fully vaccinated persons for COVID-19)21.
Data quality control: Field investigators were trained on how to conduct interview and collect data. A pre-test was done on 15 participants who were not included in the study and questions were adjusted based on any remarks/discrepancy/misunderstanding. Close supervision of data collectors was carried out by senior and supervising investigators. The internal consistency of questionnaire was determined using the Cronbachs alpha22. A Cronbach’s alpha value of 0.713 was obtained for the study questionnaire. Data collected from each participant were entered in an Excel spreadsheet, coded and double-checked for consistency completeness and missing data. Any discrepancy was resolved through discussion, consensus and discussion with senior and supervising investigators.
Statistical analysis: Statistical analysis was performed using StatView v5.0 (SAS Institute, Chicago, Inc., Illinois, United States of America), statistical package for social sciences-SPSS v16 (SPSS IBM, Inc., Chicago, Illinois, United States of America) and GraphPad v5.03 (GraphPad PRISM, San Diego, Inc., California, United States of America) software. Categorical variables were summarized as percentages with 95% confidence intervals (95% CI), while continuous variables were presented as Mean±Standard deviation (SD). Pearson’s independence Chi Square and Fisher’s Exact Tests were used to compare proportions. Univariate and multivariate logistic regression analyses were used to identify determinants of COVID-19 vaccination coverage. The association between independent variables and COVID-19 vaccination coverage was analyzed by calculating crude and adjusted values of odds ratios (cOR and aOR). These estimates were then converted into corresponding risk ratios (RR) as described by Zhang and Yu23. A p<0.05 was considered statistically significant for all analyses.
Ethical statements: Ethical clearances were issued by the institutional review boards of University of Douala (N°2945 CEI-UDo/12/2021/T) and the Littoral health regional delegation (N°0038/AAR/MINSANTE/DRSPL/BCASS). Each participant who signed a written informed consent form was ensured on the anonymisation and confidentiality of their personal information. Participation was voluntary and without financial compensation.
RESULTS
Participants included in the study: During the study period, a total of 2354 individuals were approached at the health facilities. After exclusion of 1934 individuals based on the exclusion criteria, 420 individuals agreed to participate to the study (Fig. 1).
Characteristics of participants: The study population was mainly constituted of males (52.9%), giving a male-to-female ratio sex of 1.12. Patients aged ≥60 years accounted for 16% of the participants. Most of the participants were had completed university studies (60.5%). Based on medical records and discussion with participants and medical doctors, comorbidities such as diabetes and hypertension were reported in 6.4 and 11.2%, respectively (Table 1).
Proportion of COVID-19 vaccination uptake: Of the 420 individuals included in the study, 78 individuals received at least one vaccine dose. Thus, the prevalence of COVID-19 vaccination uptake was 18.6% (95, CI 15.1-22.6%).
Type and completeness of COVID-19 vaccination: Patients were COVID-19 vaccinated with 5 types of vaccines: Johnson and Johnson, Sinopharm, Oxford/AstraZeneca, Moderna and Pfizer BioNTech. Vaccine coverage was mainly achieved with Janssen (34.6%), Sinopharm (24.4%) and Oxford/AstraZeneca (20.5%) (Table 2). Of the 78 vaccinated, 74 (94.9%) of them were fully vaccinated. Of note, six patients received two different types of COVID-19 vaccines, 4 received Moderna+Pfizer BioNTech, one received Oxford/AstraZeneca+Pfizer BioNTech and one received Pfizer BioNTech+Sinopharm.
| Table 1: | Sociodemographic, anthropometric and clinical characteristics of the participants included in the study | |||
| Characteristics | n (%) |
| Sociodemographic characteristics | |
| Male, n (%) | 222 (52.9%) |
| Age >60 years, n (%) | 68 (16.0%) |
| Mean age±SD (years) | 42.3±14.4 |
| Single, n (%) | 182 (43.3%) |
| University level, n (%) | 254 (60.5%) |
| Non-medical formal sector, n (%) | 241 (57.4%) |
| Anthropometric characteristics | |
| Weight±SD (Kg) | 78.56±15.59 |
| Height±SD (m) | 1.69±0.11 |
| BMI±SD (kg m2) | 27.54±5.14 |
| Clinical characteristics | |
| At least one comorbidity, n (%) | 161 (38.3%) |
| Obesity, n (%) | 114 (27.1%) |
| Hypertension, n (%) | 47 (11.2%) |
| Diabetes, n (%) | 27 (6.4%) |
| Asthma, n (%) | 12 (2.9%) |
| Heart failure, n (%) | 10 (2.4%) |
| HIV, n (%) | 8 (1.9%) |
| Cancer, n (%) | 3 (0.7%) |
| Stroke, n (%) | 3 (0.7%) |
| Coronary heart disease, n (%) | 1 (0.2%) |
| Renal impairment, n (%) | 1 (0.2%) |
| Gastroenteritis, n (%) | 0 (0%) |
| Gout, n (%) | 0 (0%) |
| Malaria, n (%) | 0 (0%) |
| Data are presented as frequency (n) and percentages (%) or Mean±Standard deviation (DS), BMI: Body mass index and HIV: Human immunodeficiency virus | |

|
| Table 2: | Types of COVID-19 vaccines by regard to vaccine status of the participants | |||
Fully vaccinated |
Partially vaccinated |
Total |
||||
| COVID-19 vaccines | n |
(%) |
n |
(%) |
n |
(%) |
| Janssen | 27 |
36.5 |
0 |
0 |
27 |
34.6 |
| Sinopharm | 19 |
25.6 |
0 |
0 |
19 |
24.4 |
| Oxford/AstraZeneca | 16 |
21.6 |
0 |
0 |
16 |
20.5 |
| Pfizer BioNTech | 9 |
12.2 |
0 |
0 |
9 |
11.5 |
| Moderna+Pfizer BioNTech | 2 |
2.7 |
2 |
50 |
4 |
5.1 |
| Moderna | 1 |
1.4 |
0 |
0 |
1 |
1.3 |
| Oxford/AstraZeneca+Pfizer BioNTech | 0 |
0 |
1 |
25 |
1 |
1.3 |
| Pfizer BioNTech+Sinopharm | 0 |
0 |
1 |
25 |
1 |
1.3 |
| Total | 74 |
100 |
4 |
100 |
78 |
100 |
| Data are presented as frequency (n) and percentages (%) | ||||||
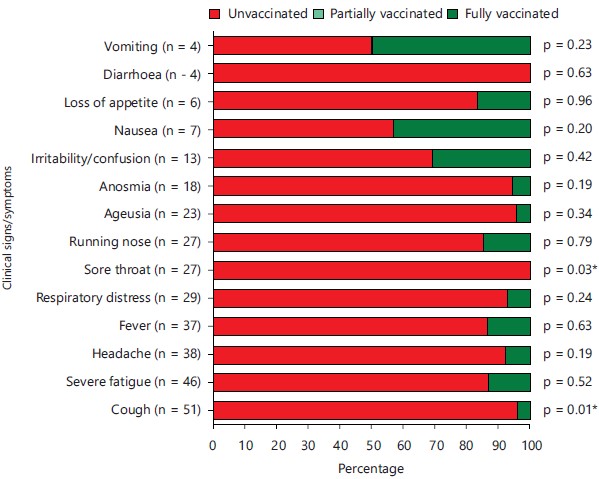
Clinical symptoms by vaccination status: Most of patients presented clinical symptoms, which were mainly represented by cough, severe fatigue and headache. Overall, prevalence of clinical symptoms was higher in unvaccinated patients, with statistically significant difference for cough (p = 0.01) and sore throat (p = 0.03). No clinical symptoms were reported in partially vaccinated patients (Fig. 2).
Prevalence of SARS-CoV-2 breakthrough infection: Of the 74 fully COVID-19 vaccinated patients, SARS-CoV-2 was detected by RT-qPCR in four patients, giving a prevalence of 5.41% (95% CI 2.12-13.1%).
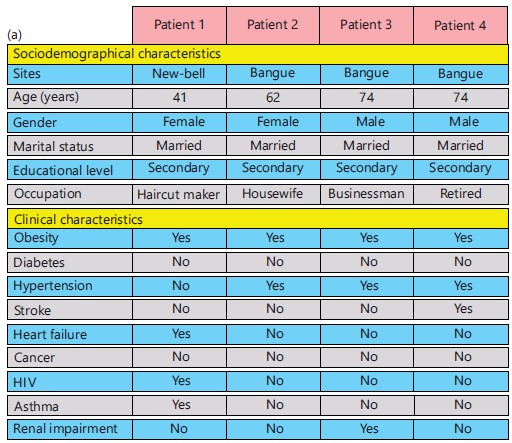
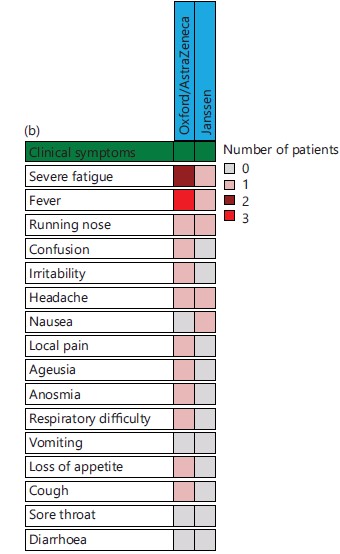
Characteristics of patients with SARS-CoV-2 breakthrough infection: On analysis of patients with SARS-CoV-2 breakthrough infection, it was noted that most of them were elderly, married and had completed secondary studies. Obesity and hypertension were main comorbidities reported in these four patients (Fig. 3a). Three of these patients were fully vaccinated with Oxford/AstraZeneca vaccine while the remaining patient was vaccinated with Janssen vaccine. A total of 13 clinical signs/symptoms were reported and were more frequently seen in those vaccinated with Oxford/AstraZeneca vaccine (Fig. 3b).
COVID-19 vaccination coverage, sociodemographic and clinical information: The distribution of COVID-19 vaccinated patients with regard to sociodemographic and clinical information is summarised in Table 3-4. The proportion of COVID-19 vaccination uptake was significantly higher in those attending site of the Bangue District Hospital (32.5%, p<0.0001), those aged >70 years old (35.3%, p = 0.02), obese (26.3%, p = 0.01) and those having history of infection (38.8%, p<0.0001).
|
| Table 3: | COVID-19 vaccination uptake and sociodemographic characteristics | |||
| Variables | Categories | N |
n (%) |
χ2 (df) |
p-value |
| Health facility | Bangue | 160 |
52 (32.5%) |
39.9 (6) |
<0.0001* |
| Boko | 21 |
2 (9.5%) |
|||
| Bonassama | 67 |
5 (7.5%) |
|||
| Cité des Palmiers | 67 |
4 (6.0%) |
|||
| Deido | 29 |
7 (24.1%) |
|||
| New-Bell | 41 |
2 (4.9%) |
|||
| Nylon | 35 |
6 (17.1%) |
|||
| Age groups (years) | <30 | 98 |
16 (16.3%) |
13.4 (5) |
0.02* |
| (30-40) | 115 |
15 (13.0%) |
|||
| (40-50) | 78 |
14 (17.9%) |
|||
| (50-60) | 61 |
10 (16.4%) |
|||
| (60-70) | 51 |
17 (33.3%) |
|||
| 70+ | 17 |
6 (35.3%) |
|||
| Gender | Females | 198 |
35 (17.7%) |
- |
0.71 |
| Males | 222 |
43 (19.4%) |
|||
| Matrimonial status | Single | 182 |
32 (17.6%) |
0.59 (2) |
0.74 |
| Married | 223 |
44 (19.7%) |
|||
| Divorced/widow (er) | 15 |
2 (13.3%) |
|||
| Level of education | None/primary | 25 |
3 (12.0%) |
1.12 (2) |
0.57 |
| Secondary | 141 |
29 (20.6%) |
|||
| University | 254 |
46 (18.1%) |
|||
| Occupation | Student | 50 |
9 (18.0%) |
0.33 (2) |
0.84 |
| Formal sector | 283 |
51 (18.0%) |
|||
| Informal sector | 87 |
18 (20.7%) |
|||
| Independence pearsons Chi-Square Test was used to compare percentages, χ2: Decision variable of Chi-Square Test, df: Degree of freedom and *Statistically significant at p<0.05 | |||||
|
Determinants of COVID-19 vaccination uptake: Based on univariate logistic regression analysis, 4 determinants were found to be associated with COVID-19 vaccination uptake namely health facility, age, obesity and history of COVID-19 infection (Table 5). The odds of getting vaccinated were reduced by 52-76% in patient attending Boko health care centre (aRR = 0.48, 95% CI 0.07-0.98, p = 0.04), Bonassama district hospital (aRR = 0.37, 95%CI 0.09-0.54, p = 0.0003), Cité des Palmiers District Hospital (aRR = 0.28, 95%CI 0.07-0.48, p = 0.002) and New Bell District Hospital (aRR = 0.24, 95%CI 0.03-0.56 and p = 0.002), compared to those attending the site of the Bangue District Hospital.
| Table 4: | COVID-19 vaccination uptake and clinical characteristics | |||
| Variables | Categories |
N |
n (%) |
χ2 (df) |
p-value |
| Obesity | No |
306 |
48 (15.7) |
- |
0.01* |
Yes |
114 |
30 (26.3) |
|||
| Diabetes | No |
393 |
72 (18.3) |
- |
0.61 |
Yes |
27 |
6 (22.2) |
|||
| Hypertension | No |
373 |
68 (18.2) |
- |
0.69 |
Yes |
47 |
10 (21.3) |
|||
| Heart failure | No |
410 |
76 (18.5) |
- |
0.91 |
Yes |
10 |
2 (20.0) |
|||
| HIV | No |
412 |
75 (18.2) |
- |
0.17 |
Yes |
8 |
3 (37.5) |
|||
| Asthma | No |
408 |
77 (18.9) |
- |
0.71 |
Yes |
12 |
1 (8.3) |
|||
| History of COVID-19 | No |
353 |
52 (14.7) |
21.6 (1) |
<0.0001* |
Yes |
67 |
26 (38.8) |
|||
| Independence pearsons Chi-Square Test was used to compare percentages, χ2: Decision variable of Chi-Square Test, df: Degree of freedom and *Statistically significant at p<0.05 | |||||
| Table 5: | Univariate logistic regression of determinants COVID-19 vaccination uptake | |||
| Variables | Categories | cOR (95%CI) | aRR (95%CI) | p-value |
| Health facility | Bangue | 1 | 1 | |
| Boko | 0.22 (0.05-0.97) | 0.48 (0.07-0.98) | 0.04* |
|
| Bonassama | 0.17 (0.06-0.44) | 0.37 (0.09-0.54) | 0.0003* |
|
| Cité des Palmiers | 0.13 (0.05-0.38) | 0.28 (0.07-0.48) | 0.002* |
|
| Deido | 0.66 (0.27-1.65) | 1.32 (0.35-1.36) | 0.37 |
|
| New-Bell | 0.11 (0.02-0.46) | 0.24 (0.03-0.56) | 0.002* |
|
| Nylon | 0.43 (0.17-1.10) | 0.90 (0.23-1.07) | 0.07 |
|
| Age groups (years) | <30 | 1 | 1 | |
| (30-40) | 0.77 (0.36-1.65) | 1.07 (0.40-1.49) | 0.49 |
|
| (40-50) | 1.12 (0.51-2.47) | 1.53 (0.55-1.99) | 0.77 |
|
| (50-60) | 1.00 (0.42-2.38) | 1.38 (0.52-1.64) | 0.99 |
|
| (60-70) | 2.56 (1.16-5.65) | 2.93 (1.13-3.21) | 0.01* |
|
| 70+ | 2.80 (1.10-8.65) | 3.08 (1.08-3.85) | 0.005* |
|
| Gender | Females | 1 | 1 | |
| Males | 1.12 (0.68-1.83) | 1.56 (0.72-1.60) | 0.65 |
|
| Matrimonial status | Single | 1 | 1 | |
| Married | 1.15 (0.70-1.91) | 1.60 (0.74-1.65) | 0.58 |
|
| Divorced/widow (er) | 0.72 (0.16-3.35) | 1.04 (0.19-2.37) | 0.67 |
|
| Level of education | None/primary | 1 | 1 | |
| Secondary | 1.90 (0.53-6.79) | 2.30 (0.56-4.01) | 0.32 |
|
| University | 1.62 (0.47-5.65) | 2.01 (0.50-3.63) | 0.44 |
|
| Occupation | Student | 1 | 1 | |
| Formal sector | 1.00 (0.46-2.19) | 1.42 (0.51-1.80) | 0.99 |
|
| Informal sector | 1.19 (0.49-2.89) | 1.66 (0.54-2.16) | 0.7 |
|
| Obesity | No | 1 | 1 | |
| Yes | 1.92 (1.14-3.22) | 2.40 (1.12-2.80) | 0.01* |
|
| Diabetes | No | 1 | 1 | |
| Yes | 1.27 (0.50-3.27) | 1.76 (0.55-2.31) | 0.61 |
|
| Hypertension | No | 1 | 1 | |
| Yes | 1.21 (0.57-2.56) | 1.69 (0.62-1.99) | 0.61 |
|
| Heart failure | No | 1 | 1 | |
| Yes | 1.10 (0.23-5.28) | 1.56 (0.27-2.95) | 0.91 |
|
| HIV | No | 1 | 1 | |
| Yes | 2.70 (0.63-11.53) | 2.97 (0.68-3.95) | 0.18 |
|
| Asthma | No | 1 | 1 | |
| Yes | 0.39 (0.05-3.07) | 0.59 (0.06-2.21) | 0.37 |
|
| History of COVID-19 | No | 1 | 1 | |
| Yes | 3.67 (2.07-6.51) | 3.60 (1.79-4.68) | <0.0001* |
|
| Univariate logistic regression analysis was used to identify factors associated with COVID-19 vaccination uptake, COVID-19: Coronavirus disease 2019, 95%CI: Confidence interval at 95%, cOR: Crude odds ratio, aRR: Adjusted risk ratio, HIV: Human immunodeficiency virus, SARS-CoV-2: Severe acute respiratory syndrome Coronavirus 2 and *Statistically significant at p<0.05 | ||||
| Table 6: | Multivariate logistic regression of determinants COVID-19 vaccination uptake | |||
| Variables | Categories | aOR (95%CI) | aRR (95%CI) | p-value |
| Sociodemographic characteristics | ||||
| Health facility | Bangue | 1 | 1 | |
| Boko | 0.21 (0.04-0.94) | 0.46 (0.06-0.96) | 0.03* |
|
| Bonassama | 0.13 (0.04-0.37) | 0.28 (0.06-0.47) | 0.0002* |
|
| Cité des Palmiers | 0.08 (0.02-0.27) | 0.18 (0.03-0.35) | <0.0001* |
|
| Deido | 0.64 (0.23-1.78) | 1.28 (0.31-1.42) | 0.38 |
|
| New-Bell | 0.07 (0.01-0.34) | 0.15 (0.01-0.43) | 0.001* |
|
| Nylon | 0.47 (0.17-1.31) | 0.98 (0.23-1.19) | 0.14 |
|
| Age groups (years) | <30 | 1 | 1 | |
| (30-40) | 0.89 (0.32-2.47) | 1.23 (0.36-1.99) | 0.82 |
|
| (40-50) | 1.42 (0.46-4.34) | 1.88 (0.50-2.81) | 0.54 |
|
| (50-60) | 1.62 (0.44-6.01) | 2.10 (0.48-3.31) | 0.46 |
|
| (60-70) | 4.17 (1.19-14.56) | 3.59 (1.15-4.54) | 0.02* |
|
| 70+ | 8.37 (1.30-54.03) | 3.27 (1.24-5.60) | 0.02* |
|
| Gender | Females | 1 | 1 | |
| Males | 1.42 (0.75-2.69) | 1.92 (0.78-2.07) | 0.27 |
|
| Matrimonial status | Single | 1 | 1 | |
| Married | 0.56 (0.27-1.17) | 0.81 (0.31-1.14) | 0.12 |
|
| Divorced/widow (er) | 0.44 (0.06-3.23) | 0.64 (0.07-2.32) | 0.42 |
|
| Level of education | None/primary | 1 | 1 | |
| Secondary | 3.19 (1.60-17.12) | 3.46 (1.49-5.83) | 0.01* |
|
| University | 2.24 (0.38-13.08) | 2.65 (1.41-5.34) | 0.03* |
|
| Occupation | Student | 1 | 1 | |
| Formal sector | 0.87 (0.28-2.68) | 1.25 (0.32-2.06) | 0.81 |
|
| Informal sector | 1.29 (0.32-5.19) | 1.78 (0.36-2.96) | 0.72 |
|
| Obesity | No | 1 | 1 | |
| Yes | 2.29 (1.21-4.71) | 2.73 (1.17-2.98) | 0.01* |
|
| Diabetes | No | 1 | 1 | |
| Yes | 1.48 (0.45-4.85) | 2.00 (0.50-2.85) | 0.51 |
|
| Hypertension | No | 1 | 1 | |
| Yes | 0.61 (0.21-1.72) | 0.90 (0.25-1.52) | 0.34 |
|
| Heart failure | No | 1 | 1 | |
| Yes | 1.04 (0.12-9.16) | 1.48 (0.14-3.65) | 0.97 |
|
| HIV | No | 1 | 1 | |
| Yes | 2.69 (0.42-17.05) | 2.96 (0.47-4.35) | 0.29 |
|
| Asthma | No | 1 | 1 | |
| Yes | 0.65 (0.07-5.93) | 0.97 (0.08-3.07) | 0.7 |
|
| History of COVID-19 | No | 1 | 1 | |
| Yes | 3.63 (1.79-7.35) | 3.59 (1.60-3.80) | 0.0003* |
|
| Multivariate logistic regression analysis was used to identify factors associated with COVID-19 vaccination uptake. COVID-19: Coronavirus disease 2019, 95% CI: Confidence interval at 95%, aOR: Adjusted odds ratio, aRR: Adjusted risk ratio, HIV: Human immunodeficiency virus, SARS-CoV-2: Severe Acute Respiratory Syndrome Coronavirus 2 and *Statistically significant at p<0.05 | ||||
Conversely, the chances of COVID-19 vaccination uptake were increased by ~three times in patients aged 60-70 years (aRR = 2.93, 95%CI 1.13-3.21, p = 0.01) and by three times in patients aged >70 years (aRR = 3.08, 95%CI 1.08-3.85, p = 0.005), compared to their counterparts aged 30 years. Likewise, obese patients had more than 2 times chances to get vaccinated (aRR = 2.40, 95%CI 1.12-2.80, p = 0.01) compared to non-obese patients. Finally, the odds of COVID-19 vaccination uptake were increased by ~4 times in patients having had COVID-19 infection (aRR = 3.60, 95%CI 1.79-4.68 and p<0.0001).
The findings of univariate logistic analysis were confirmed by those of multivariate logistic analysis, given the fact that health facility, age, obesity and history of COVID-19 infection were determinants of COVID-19 vaccination uptake. Level of education was additional determinants of COVID-19 vaccination uptake Table 6). Indeed, the chances of getting COVID-19 vaccination uptake were increased by ~4 times in those having completed secondary studies (aRR = 3.46, 95%CI 1.49-5.83 and p = 0.01) and by ~3 times in those having completed university studies (aRR = 2.65, 95%CI 1.41-5.34 and p = 0.03), compared to those with none/primary study level (Table 6).
DISCUSSION
Vaccination is a crucial public health measure to control infectious diseases. In this study, prevalence, clinical profile and determinants of COVID-19 vaccination uptake and SARS-CoV-2 breakthrough infection were determined among individuals. The overall prevalence of COVID-19 vaccination uptake was 18.6%. The chances of getting vaccinated were increased in those with higher education, elderly people and those with a history of COVID-19 infection. Vaccine breakthrough infections were found at a prevalence of 5.41% and were mainly seen in elderly patients with comorbidities (obesity, hypertension).
The COVID-19 vaccination uptake was relatively low in the study, which is lower than that reported previously in Cameroon24,25 and a global estimate of COVID-19 vaccination uptake rate reported by Zhang and colleagues15. Other studies reported higher vaccine uptake in specific groups such as elderly persons, college students and transborder populations26-28. Several factors drive the intention to accept, delay and abstain from vaccination and these include trust in vaccine approval, the perceived effectiveness of the vaccine for protecting others and conspiracy beliefs29,30. This phenomenon also known as COVID-19 vaccine hesitancy is well known in Cameroon31-33 and other African countries34.
Interestingly, few cases of SARS-CoV-2 breakthrough infections were found at prevalence of 5.41% in this study. Similar to higher breakthrough infection rates were reported recently in India (5.6 and 16.8%)35,36. This finding was the first report of such data in Cameroon and support the fact that current COVID-19 vaccines, even though highly efficient to prevent severe clinical forms of the disease, do not prevent SARS-CoV-2 transmission12,14. Most of these SARS-CoV-2 breakthrough infections were seen in elderly patients with comorbidities (obesity, hypertension). This result was consistent with that reported by Duarte et al.37 and is not surprising as advanced age and comorbidities are risk factors of severe clinical forms and adverse outcomes38. In Qatar, Butt and colleagues39 found an increased risk of severe COVID-19 and deaths in aged patients with vaccine breakthrough infection. Similar findings were also reported in Tunisia and Italy40,41. Other factors such as waning immunity, type of vaccines and SARS-CoV-2 variants (e.g., Omicron, Delta) can also modulate natural history of infection and thus shaped risk of vaccine breakthrough infection in populations42-45, especially in vulnerable groups such as elderly people, even in whom booster vaccine dose can fail to prevent breakthrough infection as recently reported46.
Advanced age was positively associated with COVID-19 vaccine uptake and this finding was reported previously30,47-49. It is now well described that elderly people are more at risk of complications and deaths due to COVID-19 infection. Thus, elderly patients or their relatives may confront the disease with more anxiety and fear, thereby resulting in an increased chances of vaccination rates in them. This perception of dangerousness towards COVID-19 among elderly patients could also explain why obese patients had more chances to get vaccinated. Zhao and colleagues reported similar finding among Chinese patients47. Obesity is also a strong risk factors of complications and mortality in COVID-19 infected patients38,50. Also, sensitisation actions implemented by Government of Cameroon, might also encouraged these patients to take the vaccine. Moreover, during COVID-19 crisis period, most of the countries have first targeted groups at risk of severe and deaths (elderly people, those with comorbidities) during vaccination campaigns. Several studies reported high vaccination rates in high-risk groups51,52.
In this study, vaccination uptake rate was increased in patients with higher level of education, which support findings from studies conducted elsewhere27,30,49,53. Higher educational level is known to be associated with better access and understanding of knowledge on dangerousness and prevention of diseases. The impact positive of higher educational level on preventive practices has been showed for other important diseases such as malaria54,55.
This study has several limitations. Firstly, other potential drivers of COVID-19 vaccine non-uptake such as interpersonal, anthropological and social factors were not investigated in the present study27,28,56 and this constitutes a main limitation of the study. Secondly, information on molecular typing of SARS-CoV-2 lineage found in fully vaccinated patients is lacking and this might have introduced interpretation bias. Thirdly, it was tricky to objectively distinct clinical signs/symptoms due to SARS-CoV-2 infection, COVID vaccine and other concurrent infections or ailments (e.g., malaria, bacterial and viral infections). Finally, this study was conducted in Douala and thus findings are not generalizable to whole Cameroonian population. Despite these limitations this study provides the first insights into prevalence of COVID-19 vaccination and SARS-Cov-2 breakthrough infection in Douala, the most populous town of Cameroon. Futhermore, this study outlines the need to develop information, education and communication strategies to improve COVID-19 vaccination coverage in young population and patients with low education.
CONCLUSION
This is the first report on prevalence and determinants of COVID-19 vaccination uptake, along with associated side effects. COVID-19 vaccine uptake rate was relatively low in the participants. Advanced age, higher level of education, health facility, presence of comorbidity (obesity) and history of COVID-19 were determinants of COVID-19 vaccination uptake. Twelve side effects were reported following COVID-19 vaccination uptake and these were mainly mild. In a context of high vaccine hesitancy in African populations, there is a need for adequately tailored information campaigns to sensitize Cameroonian populations on positive impact of COVID-19 vaccination, especially in elderly persons and those with low level of education and comorbidities.
SIGNIFICANCE STATEMENT
The COVID-19 is still an important public health concern in Cameroon, despite the arsenal of strategies deployed to control the pandemic including vaccination. This study aimed at determining prevalence, clinical profile and determinants of COVID-19 vaccination uptake and SARS-CoV-2 breakthrough infection in Douala, Cameroon. The study outlines a low vaccination coverage, with higher education, older age and past COVID-19 infection as main determinants. Patients were vaccinated mainly with Janssen, Sinopharm and Oxford/AstraZeneca vaccines. Vaccine breakthrough infections were found at a prevalence of 5.41% and were mainly seen in elderly patients with comorbidities including obesity and hypertension. This study pinpoints the need for ongoing surveillance of SARS-CoV-2 and increased vaccination coverage in Cameroon.
ACKNOWLEDGMENTS
The authors wish to thank patients having accepted to participate in the study. We also acknowledge support and technical assistance of managing authorities, medical doctors and staff of health facilities. We are also grateful to Mr. Stephane Koum (Department of Earth Sciences, Faculty of Sciences, The University of Douala, Cameroon) for generating map. We thank critical evaluation of this paper made by anonymous reviewers.
REFERENCES
- Kojom, L.P. and V. Singh, 2021. A review on emerging infectious diseases prioritized under the 2018 who research and development blueprint: Lessons from the Indian context. Vector-Borne Zoonotic Dis., 21: 149-159.
- Carabelli, A.M., T.P. Peacock, L.G. Thorne, W.T. Harvey and J. Hughes et al., 2023. SARS-CoV-2 variant biology: Immune escape, transmission and fitness. Nat. Rev. Microbiol., 21: 162-177.
- Tali, S.H.S., J.J. LeBlanc, Z. Sadiq, O.D. Oyewunmi and C. Camargo et al., 2021. Tools and techniques for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)/COVID-19 detection. Clin. Microbiol. Rev., 34.
- Gautret, P., M. Million, P.A. Jarrot, L. Camoin-Jau and P. Colson et al., 2020. Natural history of COVID-19 and therapeutic options. Expert Rev. Clin. Immunol., 16: 1159-1184.
- Bandeira, L., M. Lazaretti-Castro and N. Binkley, 2022. Clinical aspects of SARS-CoV-2 infection and vitamin D. Rev. Endocr. Metab. Disord., 23: 287-291.
- Jung, F., V. Krieger, F.T. Hufert and J.H. Küpper, 2020. How we should respond to the Coronavirus SARS-CoV-2 outbreak: A German perspective. Clin. Hemorheol. Microcirc., 74: 363-372.
- Sharma, A., I.A. Farouk and S.K. Lal, 2021. COVID-19: A review on the novel coronavirus disease evolution, transmission, detection, control and prevention. Viruses, 13.
- Watson, O.J., G. Barnsley, J. Toor, A.B. Hogan, P. Winskill and A.C. Ghani, 2022. Global impact of the first year of COVID-19 vaccination: A mathematical modelling study. Lancet Infect. Dis., 22: 1293-1302.
- Amanat, F. and F. Krammer, 2020. SARS-CoV-2 vaccines: Status report. Immunity, 52: 583-589.
- Schenten, D. and D. Bhattacharya, 2021. Immunology of SARS-CoV-2 Infections and Vaccines. In: Advances in Immunology, Alt, F.W. and K.M. Murphy (Eds.), Academic Press, Cambridge, Massachusetts, ISBN: 9780128245996, pp: 49-97.
- Raman, R., K.J. Patel and K. Ranjan, 2021. COVID-19: Unmasking emerging SARS-CoV-2 variants, vaccines and therapeutic strategies. Biomolecules, 11.
- Zeng, B., L. Gao, Q. Zhou, K. Yu and F. Sun, 2022. Effectiveness of COVID-19 vaccines against SARS-CoV-2 variants of concern: A systematic review and meta-analysis. BMC Med., 20.
- Kai, X., T. Xiao-Yan, L. Miao, L. Zhang-Wu and C. Jiang-Nan et al., 2021. Efficacy and safety of COVID-19 vaccines: A systematic review. Chin. J. Contemp. Pediatr., 23: 221-228.
- Korang, S.K., E. von Rohden, A.A. Veroniki, G. Ong and O. Ngalamika et al., 2022. Vaccines to prevent COVID-19: A living systematic review with trial sequential analysis and network meta-analysis of randomized clinical trials. PLoS ONE, 17.
- Wang, Q., S. Hu, F. Du, S. Zang and Y. Xing et al., 2022. Mapping global acceptance and uptake of COVID-19 vaccination: A systematic review and meta-analysis. Commun. Med., 2.
- Titanji, V.P.K., 2020. Priority research themes in the fight against the COVID-19 with particular reference to Cameroon. J. Cameroon Acad. Sci., 15: 209-217.
- Soubgui, A.F.M., E.L.E. Enyegue, L.P.K. Foko, W.S.N. Mboussi and G.D. Hogoue et al., 2023. Epidemiological situation of SARS-CoV-2 infection in Douala, the most populated and highly heterogeneous town of Cameroon: A post-vaccination update. Acta Trop., 241.
- Ye, Y., Q. Zhang, X. Wei, Z. Cao, H.Y. Yuan and D.D. Zeng, 2022. Equitable access to COVID-19 vaccines makes a life-saving difference to all countries. Nat. Hum. Behav., 6: 207-216.
- Foko, L.P.K., N.P. Nolla, H.N. Nyabeyeu, C. Tonga and L.G. Lehman, 2021. Prevalence, patterns, and determinants of malaria and malnutrition in Douala, Cameroon: A cross-sectional community-based study. BioMed Res. Int., 2021.
- Lwanga, S.K., S. Lemeshow and WHO, 1991. Sample Size Determination in Health Studies: A Practical Manual. World Health Organization, Geneva, Switzerland, ISBN-13: 9789241544054, Pages: 80.
- Lipsitch, M., F. Krammer, G. Regev-Yochay, Y. Lustig and R.D. Balicer, 2022. SARS-CoV-2 breakthrough infections in vaccinated individuals: Measurement, causes and impact. Nat. Rev. Immunol., 22: 57-65.
- Tavakol, M. and R. Dennick, 2011. Making sense of Cronbach's alpha. Intl. J. Med. Educ., 2: 53-55.
- Zhang, J. and K.F. Yu, 1998. A method of correcting the odds ratio in cohort studies of common outcomes. JAMA, 280: 1690-1691.
- Loteri, O., A.F.M. Sobgui, L.P.K. Foko, W.S.N. Mboussi, C.M. Sike, E.L.E. Enyegue and M.L.K. Mogtomo, 2022. COVID-19 and comorbidities in Douala, Cameroon. Int. J. Trop. Dis. Health, 43: 21-38.
- Amani, A., T. Mossus, F.Z.L. Cheuyem, C. Bilounga and P. Mikamb et al., 2022. Gender and COVID-19 vaccine disparities in Cameroon. COVID, 2: 1715-1730.
- Wotring, A.J., M. Hutchins, M.K. Johnson, S.F. Ferng and C. Strawser et al., 2022. COVID-19 vaccine uptake among college students at a Midwest University. J. Community Health, 47: 292-297.
- Bojorquez, I., R. Leyva-Flores, C. Rodríguez-Chávez, C. Hernández-Campos and M. Arévalo et al., 2022. Determinants of COVID-19 vaccine acceptance and uptake in a transborder population at the Mexico-Guatemala Border, September-November 2021. Int. J. Environ. Res. Public Health, 19.
- Jung, Y.J., A. Gagneux-Brunon, M. Bonneton, E. Botelho-Nevers, P. Verger, J.K. Ward and O. Launay, 2022. Factors associated with COVID-19 vaccine uptake among French population aged 65 years and older: Results from a national online survey. BMC Geriatrics, 22.
- Burke, P.F., D. Masters and G. Massey, 2021. Enablers and barriers to COVID-19 vaccine uptake: An international study of perceptions and intentions. Vaccine, 39: 5116-5128.
- Galanis, P., I. Vraka, O. Siskou, O. Konstantakopoulou, A. Katsiroumpa, I. Moisoglou and D. Kaitelidou, 2022. Cross-sectional assessment of predictors for COVID-19 vaccine uptake: An online survey in Greece. Vacunas, 23: S60-S66.
- Dinga, J.N., A.A. Njoh, S.D. Gamua, S.E. Muki and V.P.K. Titanji, 2022. Factors driving Covid-19 vaccine hesitancy in Cameroon and their implications for Africa: A comparison of two cross-sectional studies conducted 19 months apart in 2020 and 2022. Vaccines, 10.
- Yakum, M.N., A.D. Funwie, A.B. Ajong, M. Tsafack, L.E.E. Ze and Z. Shah, 2022. The burden of vaccine hesitancy for routine immunization in Yaounde-Cameroon: A cross-sectional study. PLOS Global Public Health, 2.
- Ajonina-Ekoti, I.U., K.B. Ware, C.K. Nfor, E.A. Akomoneh and A. Djam et al., 2022. COVID-19 perceptions and vaccine hesitancy: Acceptance, attitude, and barriers among Cameroonians. J. Am. Pharm. Assoc., 62: 1823-1829.
- Ackah, B.B.B., M. Woo, L. Stallwood, Z.A. Fazal, A. Okpani, U.V. Ukah and P.A. Adu, 2022. COVID-19 vaccine hesitancy in Africa: A scoping review. Global Health Res. Policy, 7.
- Doke, P.P., S.T. Mhaske, G. Oka, R. Kulkarni, V. Muley, A.C. Mishra and V.A. Arankalle, 2022. SARS-CoV-2 breakthrough infections during the second wave of COVID-19 at Pune, India. Front. Public Health, 10.
- Dhumal, S., A. Patil, A. More, S. Kamtalwar and A. Joshi et al., 2022. SARS-CoV-2 reinfection after previous infection and vaccine breakthrough infection through the second wave of pandemic in India: An observational study. Int. J. Infect. Dis., 118: 95-103.
- Duarte, L.F., N.M.S. Gálvez, C. Iturriaga, F. Melo-González and J.A. Soto et al., 2021. Immune profile and clinical outcome of breakthrough cases after vaccination with an inactivated SARS-CoV-2 vaccine. Front. Immunol., 12.
- Kompaniyets, L., A.F. Pennington, A.B. Goodman, H.G. Rosenblum and B. Belay et al., 2021. Underlying medical conditions and severe illness among 540,667 adults hospitalized with COVID-19, March 2020-March 2021. Preventing Chronic Dis., 18.
- Butt, A.A., H. Nafady-Hego, H. Chemaitelly, A.B. Abou-Samra and A. Al Khal et al., 2021. Outcomes among patients with breakthrough SARS-CoV-2 infection after vaccination. Int. J. Infect. Dis., 110: 353-358.
- Fredj, S.B., R. Ghammem, N. Zammit, A. Maatouk and N. Haddad et al., 2022. Risk factors for severe Covid-19 breakthrough infections: An observational longitudinal study. BMC Infect. Dis., 22.
- Basso, P., C. Negro, L. Cegolon and F.L. Filon, 2022. Risk of vaccine breakthrough SARS-CoV-2 infection and associated factors in healthcare workers of trieste teaching hospitals (North-Eastern Italy). Viruses, 14.
- Chen, Y., C. Li, F. Liu, Z. Ye and W. Song et al., 2022. Age-associated SARS-CoV-2 breakthrough infection and changes in immune response in a mouse model. Emerging Microb. Infect., 11: 368-383.
- Hacisuleyman, E., C. Hale, Y. Saito, N.E. Blachere and M. Bergh et al., 2021. Vaccine breakthrough infections with SARS-CoV-2 variants. New Engl. J. Med., 384: 2212-2218.
- Feikin, D.R., M.M. Higdon, L.J. Abu-Raddad, N. Andrews and R. Araos et al., 2022. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: Results of a systematic review and meta-regression. Lancet, 399: 924-944.
- Zhang, M., Y. Liang, D. Yu, B. Du and W. Cheng et al., 2022. A systematic review of vaccine breakthrough infections by SARS-CoV-2 delta variant. Int. J. Biol. Sci., 18: 889-900.
- Kuhlmann, C., C.K. Mayer, M. Claassen, T. Maponga and W.A. Burgers et al., 2022. Breakthrough infections with SARS-CoV-2 omicron despite mRNA vaccine booster dose. Lancet, 399: 625-626.
- Zhao, H., H. Wang, H. Li, W. Zheng and T. Yuan et al., 2021. Uptake and adverse reactions of COVID-19 vaccination among people living with HIV in China: A case-control study. Hum. Vaccines Immunother., 17: 4964-4970.
- Moisoglou, I., C. Passali, M. Tsiachri and P. Galanis, 2023. Predictors of COVID-19 vaccine uptake in teachers: An on-line survey in Greece. J. Community Health, 48: 59-66.
- Figueiredo, J.C., U. Ihenacho, N.M. Merin, O. Hamid and J. Darrah et al., 2022. SARS-CoV-2 vaccine uptake, perspectives, and adverse reactions following vaccination in patients with cancer undergoing treatment. Ann. Oncol., 33: 109-111.
- Frühbeck, G., J.L. Baker, L. Busetto, D. Dicker and G.H. Goossens et al., 2020. European association for the study of obesity position statement on the global COVID-19 pandemic. Obes. Facts, 13: 292-296.
- Selim, R., J. Wellens, L. Marlow and J.J. Satsangi, 2021. SARS-CoV-2 vaccination uptake by patients with inflammatory bowel disease on biological therapy. Lancet Gastroenterol. Hepatol., 6.
- Widdifield, J., L. Eder, S. Chen, J.C. Kwong and C. Hitchon et al., 2022. COVID-19 vaccination uptake among individuals with immune-mediated inflammatory diseases in Ontario, Canada, between December 2020 and October 2021: A population-based analysis. J. Rheumatol., 49: 531-536.
- Więsik-Szewczyk, E., M. Ziętkiewicz, A. Będzichowska, K. Napiórkowska-Baran, A. Matyja-Bednarczyk, A. Felis-Giemza and K. Jahnz-Różyk, 2022. Coronavirus disease 2019 vaccination uptake and hesitancy among Polish patients with inborn errors of immunity, autoinflammatory syndromes, and rheumatic diseases: A multicenter survey. Front. Immunol., 13.
- Nchetnkou, C.M., L.P.K. Foko and L.G. Lehman, 2020. Knowledge, attitude, and practices towards malaria among employees from enterprises in the Town of Douala, Cameroon. BioMed Res. Int., 2020.
- Kojom, L.P.F. and L.G. Lehman, 2018. Knowledge and beliefs towards malaria and associated factors among residents of the town of Douala, Cameroon. Arch. Curr. Res. Int., 14.
- Sumerlin, T.S., J.H. Kim, Z. Wang, A.Y.K. Hui and R.Y. Chung, 2022. Determinants of COVID-19 vaccine uptake among female foreign domestic workers in Hong Kong: A cross-sectional quantitative survey. Int. J. Environ. Res. Public Health, 19.
How to Cite this paper?
APA-7 Style
Moguem Soubgui,
A.F., Foko,
L.P., Embolo Enyegue,
E.L., Mboussi,
W.S., Koanga Mogtomo,
M. (2023). Prevalence, Clinical Profile and Determinants of COVID-19 Vaccination and SARS-CoV-2 Breakthrough Infection in Douala, Cameroon. Trends in Medical Research, 18(1), 122-135. https://doi.org/10.3923/tmr.2023.122.135
ACS Style
Moguem Soubgui,
A.F.; Foko,
L.P.; Embolo Enyegue,
E.L.; Mboussi,
W.S.; Koanga Mogtomo,
M. Prevalence, Clinical Profile and Determinants of COVID-19 Vaccination and SARS-CoV-2 Breakthrough Infection in Douala, Cameroon. Trends Med. Res 2023, 18, 122-135. https://doi.org/10.3923/tmr.2023.122.135
AMA Style
Moguem Soubgui
AF, Foko
LP, Embolo Enyegue
EL, Mboussi
WS, Koanga Mogtomo
M. Prevalence, Clinical Profile and Determinants of COVID-19 Vaccination and SARS-CoV-2 Breakthrough Infection in Douala, Cameroon. Trends in Medical Research. 2023; 18(1): 122-135. https://doi.org/10.3923/tmr.2023.122.135
Chicago/Turabian Style
Moguem Soubgui, Arlette, Flore, Loick Pradel Kojom Foko, Elisée Libert Embolo Enyegue, Wilfried Steve Ndeme Mboussi, and Martin Luther Koanga Mogtomo.
2023. "Prevalence, Clinical Profile and Determinants of COVID-19 Vaccination and SARS-CoV-2 Breakthrough Infection in Douala, Cameroon" Trends in Medical Research 18, no. 1: 122-135. https://doi.org/10.3923/tmr.2023.122.135

This work is licensed under a Creative Commons Attribution 4.0 International License.